Synergistic Zn/Cu Co-doping engineering for concurrent optimization of carrier transport and lattice thermal conductivity in p-type Mg3Sb2
Abstract
p-Type Mg3Sb2 possesses strong thermoelectric potential, yet effective strategies to further enhance its performance remain underexplored. In this study, we investigated the p-type Zintl-phase compound Mg3Sb2 and proposed a Zn/Cu co-doping strategy to synergistically optimize carrier transport and lattice thermal conductivity. Mg3.1-xZnxSb2 (x = 0, 0.4, 0.6, and 0.8) and Mg2.3-yZn0.8CuySb2 (y = 0, 0.075, 0.100, and 0.125) series samples were prepared via high-energy ball milling followed by hot pressing. First-principles calculations reveal that substituting Mg sites with Zn and Cu induces pronounced band-structure modulation, shifting the Fermi level into the valence band and narrowing the bandgap. These effects collectively increase hole concentration and enhance electrical conductivity. Meanwhile, the mass fluctuation and local lattice distortion introduced by co-doping intensify phonon scattering, resulting in a substantial reduction in lattice thermal conductivity. Experimentally, Zn/Cu co-doping delivers a well-balanced optimization of thermoelectric transport properties. The Mg2.2Zn0.8Cu0.1Sb2 sample achieves a power factor of 351.99 μW cm-1 K-2 and a peak figure of merit (ZT) of 0.42 at 735 K, corresponding to a 147% improvement compared with the undoped sample. This work elucidates the synergistic effects of Zn/Cu co-doping in electronic band engineering and phonon modulation, offering a promising strategy for the rational design of high-performance p-type Mg3Sb2 and other Zintl-phase thermoelectric materials.
Keywords
INTRODUCTION
Thermoelectric materials can directly convert heat into electricity, and vice versa, under a temperature gradient. Owing to their solid-state nature, silent operation, and minimal maintenance requirements, they hold great promise for applications such as waste-heat recovery and distributed energy generation[1-6]. Their performance is typically evaluated using the dimensionless figure of merit (ZT) = S2σT/κ, where S is the Seebeck coefficient, σ is the electrical conductivity, κ is the total thermal conductivity (κ = κele + κlatt; κele is the electronic thermal conductivity, and κlatt is the lattice thermal conductivity), and T is the absolute temperature[7,8]. Achieving a high ZT requires the coordinated optimization of a high power factor (PF = S2σ) together with a low κlatt[9]. However, these parameters are intrinsically interdependent, making it particularly challenging to simultaneously enhance all thermoelectric properties[10,11].
Among various thermoelectric systems, Zintl-phase compounds with the general formula AB2Sb2 (A = Mg, Yb, Ca, Ba; B = Mg, Zn, Cd, Ga, Al, etc.) have emerged as a research hotspot in recent years owing to their complex crystal structures and excellent chemical tunability[12-14]. The Zintl-phase compound Mg3Sb2 is composed of earth-abundant and low-toxicity elements, exhibits intrinsically low κ, and maintains good thermal stability. These features make it a promising environmentally friendly thermoelectric material for medium-temperature applications (500-900 K)[15-17]. Notably, n-type Mg3Sb2 has achieved a peak ZT exceeding 1.5, attributed to its high conduction-band degeneracy (Nv ≈ 6) and strong lattice anharmonicity[12,18-23]. In contrast, p-type Mg3Sb2 suffers from low valence-band degeneracy (Nv ≈ 1) and a large valence-band effective mass[12,13], which collectively restrict carrier transport, resulting in a peak ZT of less than 0.7 to date[15,24-26]. For the construction of all-magnesium-based thermoelectric devices, the relatively poor performance of p-type components remains a critical bottleneck that limits their compatibility with high-performance n-type counterparts. Therefore, developing effective strategies such as band-structure engineering and defect modulation to enhance the thermoelectric performance of p-type Mg3Sb2 represents a key challenge in this field[27].
A variety of strategies have been explored to overcome the performance bottleneck of p-type Mg3Sb2. Alkali-metal doping, such as Na[15,28] or Li[28,29], can effectively introduce holes, increase carrier concentration (n), and improve σ; however, it often leads to an increase in κlatt, which offsets part of the performance gain. Ag doping[30,31] can simultaneously increase mobility (μ) and n by reducing chemical bond polarity, although its ability to suppress κ remains limited. Electron-type dopants such as Zn[24,32] can introduce mass fluctuation and local strain fields at Mg sites, thereby enhancing phonon scattering and significantly lowering κlatt. Some studies also suggest that Zn may modify the valence-band structure and improve the PF[33]. For example, Ren et al.[33] demonstrated that Zn/Na co-doping can simultaneously suppress κlatt and enhance μ, resulting in an increase in peak ZT from 0.6 to 0.8 and a 76% improvement in the engineering performance index ZTeng. More complex co-doping strategies such as Zn/Ag[30] or Zn/Yb[12] have also yielded promising results: by introducing multiple point defects and enabling band modulation, these approaches enhance PF while reducing κ, achieving peak ZT values up to 0.84. After further Zn/Yb co-doping and carrier optimization, a peak ZT exceeding 1.1 has even been reported. Despite these advances, two major challenges remain: (i) bipolar conduction often emerges at high temperatures, causing substantial performance degradation; and (ii) the functional roles of dopants are difficult to decouple, making it challenging to simultaneously maintain high electrical performance and low κ across the full temperature range.
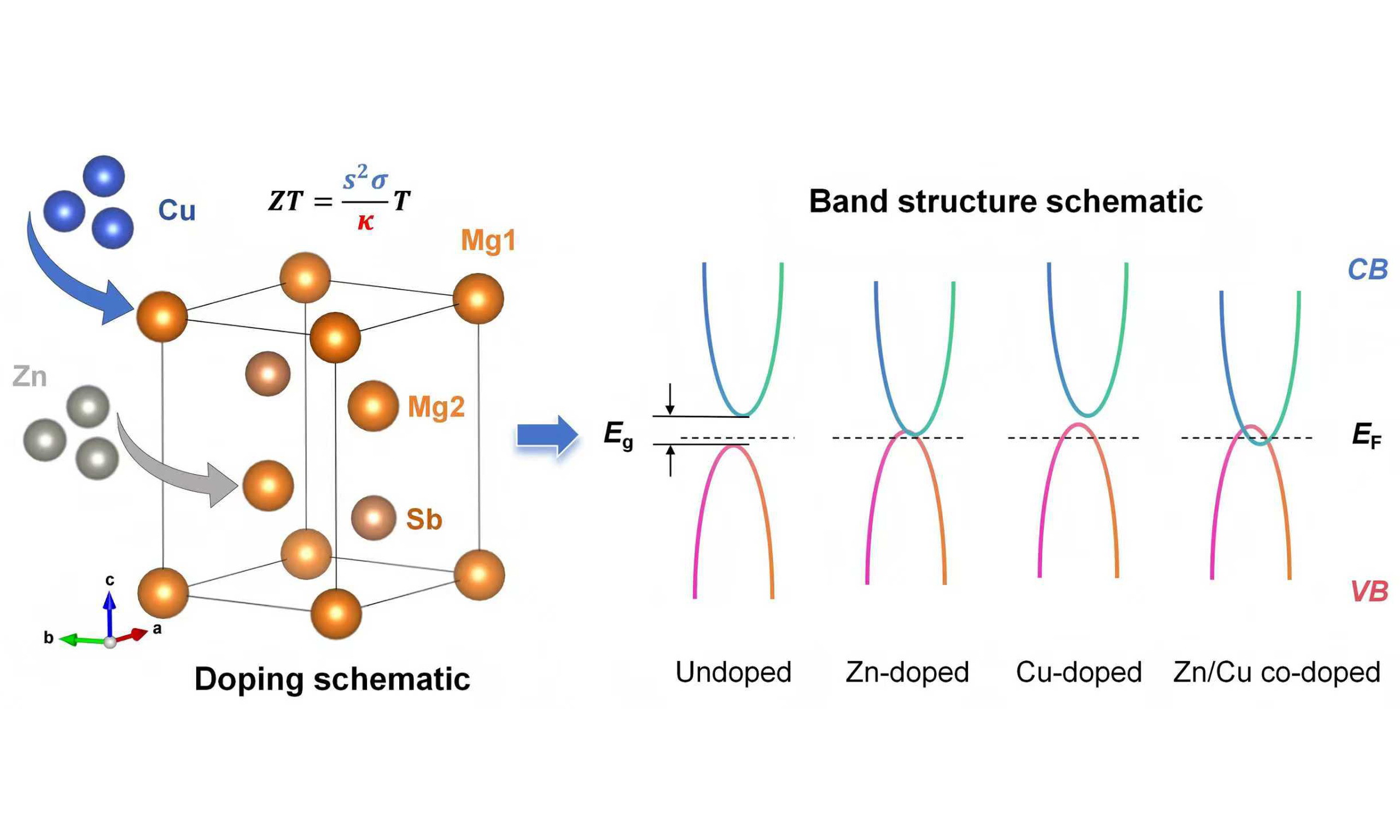
Building on the above insights, this study proposes a Zn/Cu co-doping strategy to achieve synergistic optimization of p-type Mg3Sb2-based materials, with both dopants substituting at the Mg sites. First, Zn is incorporated to introduce significant mass fluctuation and local lattice strain, which effectively scatters long-wavelength phonons and reduces the κlatt. Meanwhile, Zn incorporation increases the density of states near the Fermi level (EF), enabling modulation of the bandgap (Eg) and valence-band structure[24]. Subsequently, Cu is introduced to further tailor the valence-band edge. Benefiting from the +1 valence state of the group-IB Cu element at elevated temperatures, Cu doping modifies the local chemical environment, optimizes hole concentration, and enhances the PF. By precisely controlling the doping sequence and site occupancy of Zn and Cu, this approach aims to achieve balanced thermoelectric transport properties across the entire operating temperature range, thereby advancing the practical application of this environmentally benign Zintl-phase material.
EXPERIMENTAL
Materials synthesis
All sample preparation was conducted in an argon-filled glovebox with oxygen and moisture levels below 0.1 ppm. High-purity magnesium powder (Mg, 99.95%, Macklin), antimony powder (Sb, 99.99%, Macklin), zinc powder (Zn, 99.99%, Aladdin), and copper powder (Cu, 99.99%, Macklin) were weighed according to the nominal compositions of Mg3.1-xZnxSb2 (x = 0, 0.4, 0.6, and 0.8) and Mg2.3-yZn0.8CuySb2 (y = 0, 0.075, 0.100, and 0.125). Appropriate amounts of stainless-steel milling balls were added at a ball-to-powder mass ratio of 10:1. The mixtures were homogenized, loaded into stainless-steel milling jars, and sealed under argon. High-energy ball milling was performed for 8 h at 1,200 revolutions per minute (rpm) using a planetary mill (MSK-SFM-3, Hefei Kejing Materials Technology Co., Ltd., China). After milling, the powders were lightly ground and packed into a 12.7 mm-diameter graphite die lined with graphite foil. Densification was carried out using a hot-press furnace (ZT-40-20Y, Shanghai Chenhua Electric Furnace Co., Ltd., China) at 873 K under 60 MPa for 2 minutes. The resulting dense pellets were subsequently ground, polished, and cut into appropriate dimensions for further characterization.
Structural characterization
X-ray diffraction (XRD, D8 ADVANCE A25, Bruker, Germany) was employed to analyze the phase composition and crystal structure of the samples. Measurements were conducted using Cu Kα radiation with a step size of 0.02° and a counting time of 0.2 s per step. The fresh fracture surfaces of Mg3.1-xZnxSb2 (x = 0, 0.4, 0.6, and 0.8) and Mg2.3-yZn0.8CuySb2 (y = 0, 0.075, 0.100, and 0.125) were examined using scanning electron microscopy (SEM, FlexSEM 1000 II, Hitachi, Japan) equipped with an energy-dispersive X-ray spectrometer (EDS) to evaluate the microstructure, surface morphology, and elemental distribution.
Performance test
The S and σ were measured in a helium atmosphere using a four-probe testing system (CTA-3, Beijing Keruiou Technology Co., Ltd.). Measurements were conducted over the temperature range of 324-735 K. The κ was calculated using κ = DρCp, where the thermal diffusivity (D) was measured using a laser flash apparatus (LFA 467 HT, NETZSCH, Germany). The specific heat capacity (Cp) was estimated using the Dulong-Petit law[34]; and the sample density (ρ) was determined by the Archimedes method. The Hall coefficient (RH) was measured using the van der Pauw method under a magnetic field of 1.5 T. The n and μ were calculated using n = 1/(eRH) and μ = σRH, respectively, where e is the elementary charge. In this study, a series of precise measurements revealed that the σ fluctuates by approximately ±10% around its average value. These variations primarily arise from subtle differences in the material preparation process, limitations in instrument accuracy, and minor fluctuations in environmental conditions. In contrast, the S exhibits much higher stability, with a fluctuation of only about ±5%, demonstrating the high precision and excellent repeatability of this measurement. Similarly, the κ shows a fluctuation range of approximately ±5%, further confirming the reliability and consistency of the measurement procedure. Other key performance indicators, such as ZT and the average dimensionless figure of merit ZTave, are subsequently calculated from these three core parameters using the corresponding formulas.
Density functional theory calculations
Density functional theory (DFT) calculations were performed using the Vienna Ab initio Simulation Package (VASP) with the projector augmented-wave (PAW) method[35-37]. The generalized gradient approximation (GGA) with the fully relativistic Perdew-Burke-Ernzerhof (PBE) functional was employed to treat exchange-correlation interactions[38-40]. To model Zn and Cu doping, quasi-random alloy structures were generated using the Universal Structure Predictor: Evolutionary Xtallography (USPEX) evolutionary algorithm, which incorporates local optimization, real-space representation, and multiple physically guided mutation operators. The target dopant distribution was obtained by minimizing the structural order of a 2 × 2 × 2 supercell containing 40 atoms[17]. During geometric optimization, all atomic positions were allowed to relax freely. Brillouin-zone sampling was carried out using a Monkhorst-Pack k-point mesh with a spacing of 0.03 Å-3. The valence wavefunctions were expanded using a plane-wave basis set with a cutoff energy of
RESULTS AND DISCUSSION
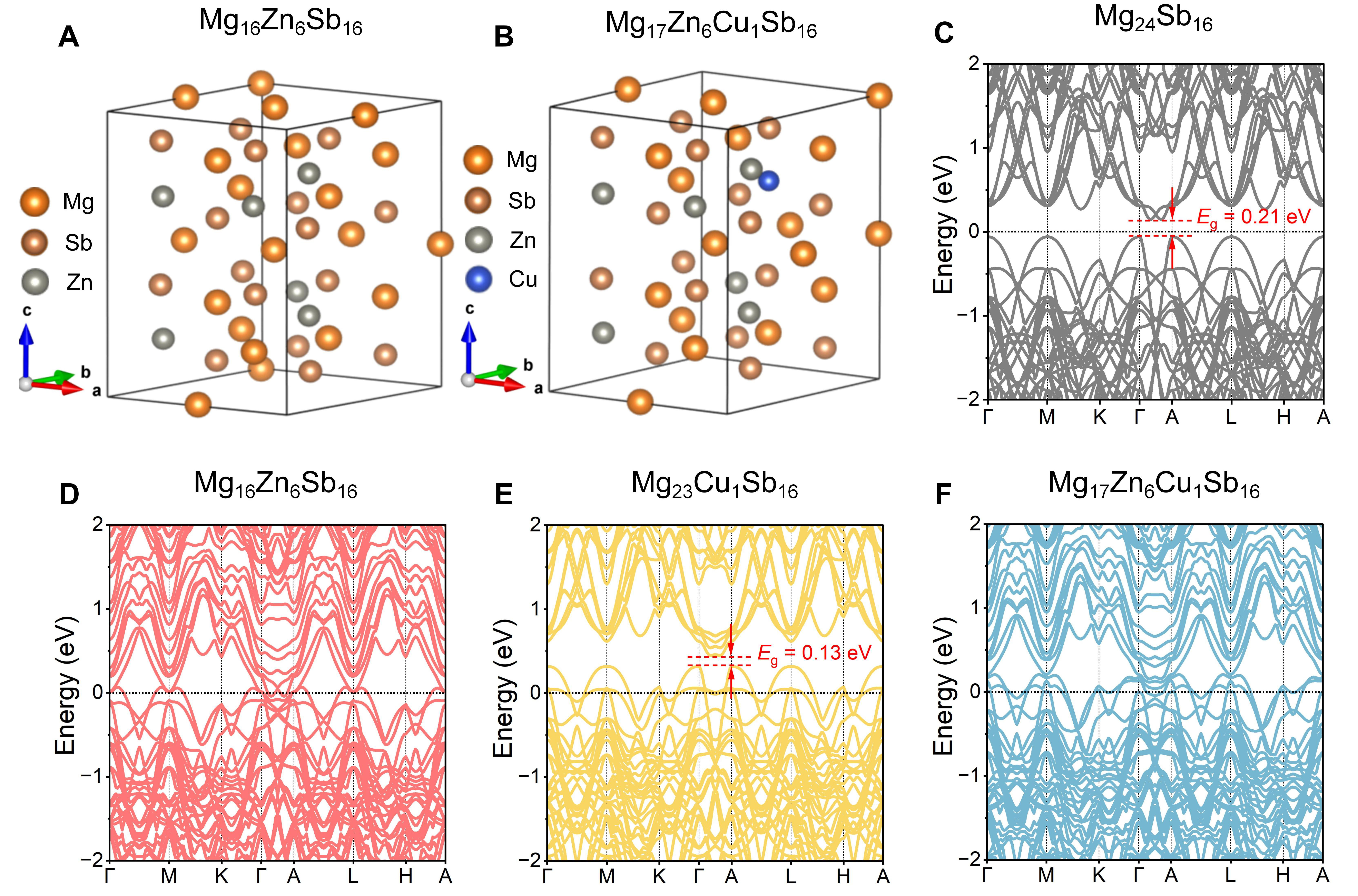
To verify the feasibility of Mg-site doping with Zn and Cu, first-principles calculations based on DFT were carried out. The supercell structures of Mg16Zn6Sb16 and Mg17Zn6Cu1Sb16 are shown in Figure 1A and B, respectively. Both exhibit the typical cage-like framework of anti-La2O3-type layered Zintl-phase compounds (space group
Figure 1. Crystal structures and electronic band structures of Mg3Sb2-based materials before and after doping. Supercell models of (A) Mg16Zn6Sb16 and (B) Mg17Zn6Cu1Sb16; calculated electronic band structures of (C) Mg24Sb16, (D) Mg16Zn6Sb16, (E) Mg23Cu1Sb16, and (F) Mg17Zn6Cu1Sb16. Eg: denotes the bandgap.
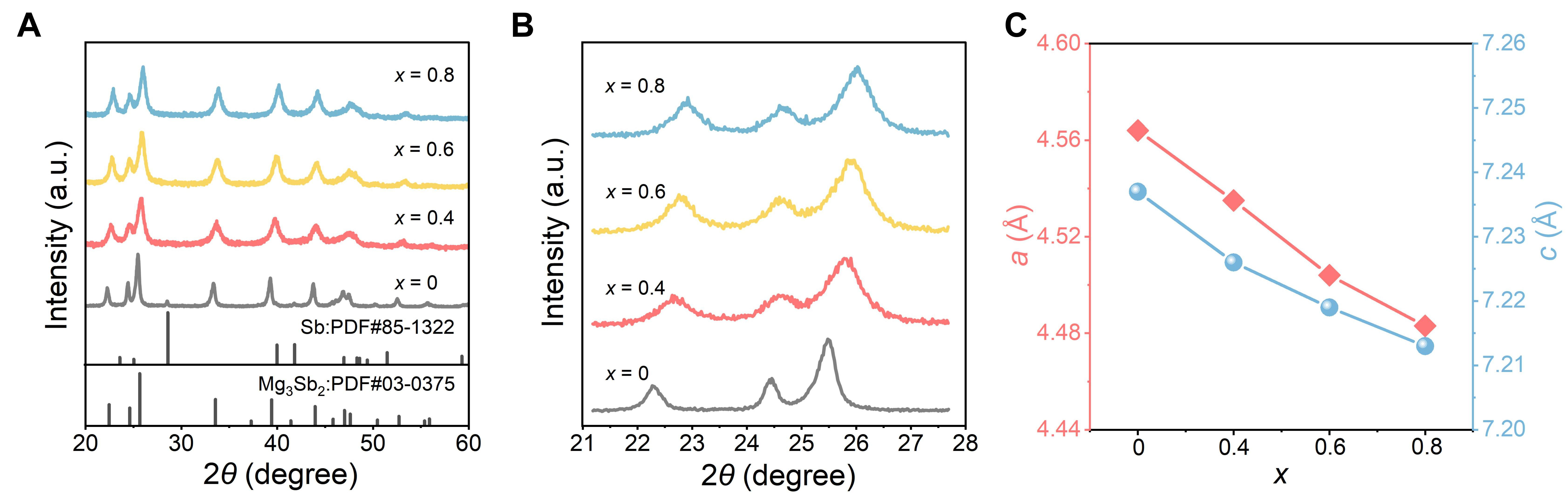
The XRD patterns of the Mg3.1-xZnxSb2 (x = 0, 0.4, 0.6, and 0.8) samples are shown in Figure 2A. Most diffraction peaks for all compositions match well with the standard PDF card of Mg3Sb2 (PDF#03-0375). A small peak at approximately 28.54° is observed only in the x = 0 sample, corresponding to trace Sb, which may originate from Mg volatilization during ball milling or sintering. The minor presence of the Sb phase may pin EF near the p-type region, which is favorable for p-type behavior[13,44]. Phase analysis was carried out using Jade 6.0 software (Materials Data, Inc., Livermore, USA). As the Zn content increases (x > 0.4), this impurity peak gradually disappears, indicating that Zn atoms successfully substitute the Mg site and stabilize the crystal structure. This observation is consistent with the findings of Ren et al.[33]. Since Zn has a higher electronegativity (1.65) than Mg (1.31), the Zn-Sb bond exhibits stronger covalency compared to the Mg-Sb bond. The introduction of Zn can optimize the unsaturated bonding environment caused by Mg deficiency, thereby suppressing Mg volatilization. During the increase of x, the enlarged view of the XRD pattern [Figure 2B] reveals that the main diffraction peaks shift from 25.49° to 26.05°, accompanied by a decrease in the lattice parameters a and c [Figure 2C]. This peak shift can be attributed to the smaller ionic radius of Zn relative to Mg in this chemical environment, leading to lattice contraction and a reduced interplanar spacing (d) upon substitution. According to Bragg’s law (2d·sinθ = nλ), a decrease in d results in an increase in θ, causing the diffraction peaks to shift toward higher angles. Similar peak-shift behavior under heavy Zn doping has been reported by Huang et al.[30], Ren et al.[33], Xin et al.[45], and Ahmadpour et al.[46]. In addition, the introduction of microstrain and point defects during solid-solution formation leads to peak broadening[47].
Figure 2. Phase characterization of Mg3.1-xZnxSb2 (x = 0, 0.4, 0.6, and 0.8). (A) X-ray diffraction (XRD) patterns in the 2θ range of 20°-60°; (B) magnified XRD patterns from 21.19° to 27.69°; (C) evolution of lattice parameters a and c with Zn content. PDF: Powder diffraction file.
Figure 3A-D illustrates the morphological evolution of the Mg3.1-xZnxSb2 (x = 0, 0.4, 0.6, and 0.8) samples. For x = 0 and x = 0.4, the sample surfaces appear relatively dense, with tightly bonded grains and only a few irregular pores. However, as the Zn content increases further (x = 0.6 and 0.8), the SEM images reveal a significant rise in both the number and size of pores in the heavily doped samples, accompanied by looser grain boundaries. This observation aligns with the measured decrease in ρ shown in [Supplementary Table 1]. We attribute this phenomenon primarily to the preparation process. For Mg-containing Zintl phases, prolonged high-energy ball milling typically leads to Mg volatilization. Additionally, as the Zn concentration increases, alloying effects may induce lattice distortions. During hot pressing, such distortions can disrupt mass transport and reduce effective diffusivity, thereby retarding interparticle neck growth. Furthermore, the extended hot-pressing duration, necessitated by the equipment’s slow heating and cooling rates
Figure 3. Microstructural characterization of Mg3.1-xZnxSb2 (x = 0, 0.4, 0.6, and 0.8). (A-D) Scanning electron microscopy (SEM) images for x = 0, 0.4, 0.6, and 0.8; (E-G) Energy-dispersive X-ray spectrometer (EDS) elemental mapping for the x = 0.6 sample; (H-J) EDS elemental mapping for the x = 0.8 sample.
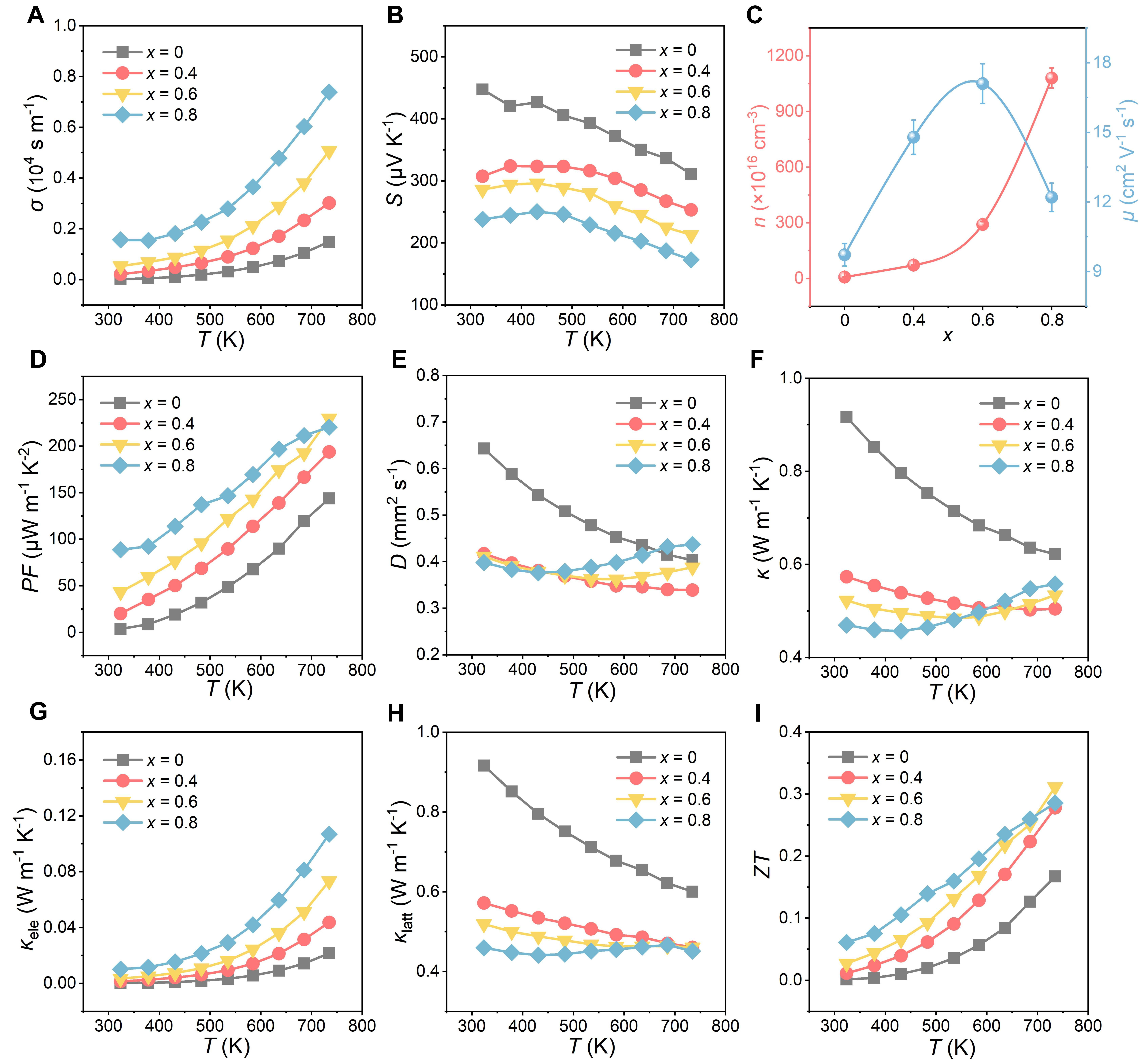
As shown in Figure 4A-I, heavy Zn doping at the Mg site significantly modifies the thermoelectric properties of Mg3Sb2-based materials. With increasing Zn content, the σ exhibits a pronounced upward trend across the entire temperature range up to 735 K [Figure 4A]. At low temperatures, limited thermal excitation results in fewer carriers and thus lower σ. As temperature increases, more holes are thermally activated into the valence band, leading to a substantial rise in n, consistent with the Hall measurements [Figure 4C]. However, heavy Zn doping shifts the EF upward, driving the system toward metallic behavior. This causes the carrier energy distribution to concentrate near EF, reducing the energy difference between the average carrier energy and EF. Consequently, the thermoelectric voltage decreases, leading to a reduction in the S [Figure 4B]. This inverse relationship between n and S is consistent with classical thermoelectric transport theory[10]. Notably, μ does not change monotonically after Zn doping but instead increases initially before decreasing [Figure 4C]. This behavior likely arises from the competing influences of point-defect scattering and grain-boundary scattering[10]. Despite the reduction in S, the PF remains significantly higher than that of the undoped sample due to the substantial increase in σ [Figure 4D]. For example, at x = 0.6, the PF reaches 230 μW cm-1 K-2 at
Figure 4. Thermoelectric properties of Mg3.1-xZnxSb₂ (x = 0, 0.4, 0.6, and 0.8). Temperature-dependent (A) electrical conductivity (σ) and (B) Seebeck coefficient (S); x-dependent (C) carrier concentration (n) and mobility (μ) at room temperature; Temperature-dependent (D) power factor (PF); (E) thermal diffusivity (D); (F) total thermal conductivity (κ); (G) electronic thermal conductivity (κele); (H) lattice thermal conductivity (κlatt), and (I) dimensionless figure of merit (ZT).
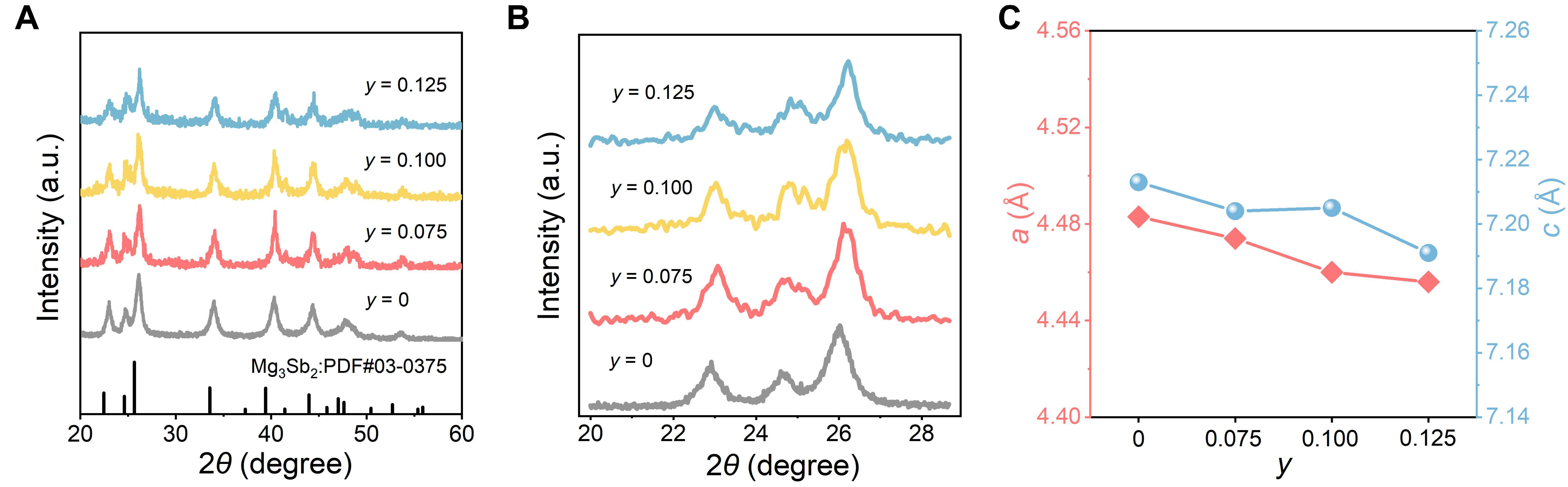
The performance enhancement achieved through single-element doping in Mg3.1Sb2 is limited; therefore, Cu was introduced into the Mg2.3Zn0.8Sb2 matrix to further improve the thermoelectric properties. Cu, as a group-IB dopant, is expected to introduce additional holes, increase the n, and further modulate the κlatt through mass fluctuation. The composition x = 0.8 was selected as the base material because it exhibits a relatively high ZTave = 0.17 over the measured temperature range, providing a more representative evaluation of temperature-dependent transport behavior [Supplementary Figure 2]. Figure 5A presents the XRD patterns of Mg2.3-yZn0.8CuySb2 (y = 0, 0.075, 0.100, and 0.125). All diffraction peaks match well with the standard Mg3Sb2 pattern (PDF#03-0375), and no detectable secondary phases are observed, indicating that the main Mg3Sb2 crystal structure is preserved across all doping levels. Figure 5B presents the magnified local XRD patterns. As the Cu content increases, the primary diffraction peaks shift slightly toward higher angles (from 26.03° to 26.20°). This peak shift indicates that Cu successfully substitutes for Mg and leads to lattice distortion. Correspondingly, the lattice parameters a and c decrease [Figure 5C], confirming a slight lattice contraction, consistent with a reduced interplanar spacing (d) and an increase in the diffraction angle θ.
Figure 5. Phase characterization of Mg2.3-yZn0.8CuySb2 (y = 0, 0.075, 0.100, and 0.125). (A) XRD patterns in the 2θ range of 20°-60°; (B) magnified XRD patterns from 20.00° to 28.68°; (C) evolution of lattice parameters a and c with increasing Cu content. XRD: X-ray diffraction.
Figure 6A-C shows the SEM images of Mg2.3-yZn0.8CuySb2 (y = 0.075, 0.100, and 0.125). Numerous small cracks and pores are visible on the fracture surfaces of samples with different Cu contents, which is consistent with the density measurement results [Supplementary Table 2]. This morphology arises partly because the heavily Zn-doped matrix dominates the overall microstructure, making the samples exhibit a similar appearance, and partly due to Mg volatilization during ball milling and hot pressing, which reduces surface densification during alloy formation. Figure 6D-G presents the EDS elemental mapping of the y = 0.100 sample. The Mg, Sb, and Zn elements are uniformly distributed across the examined area. In contrast, the Cu signal is weak overall, with localized regions of enrichment, reflecting its low doping concentration. During high-temperature synthesis or sintering, Cu may migrate and segregate along grain boundaries, leading to local concentrations that exceed the nominal average. These Cu-rich regions can act as weak scattering centers, contributing to localized modulation of σ or κ.
Figure 6. Microstructure of Mg2.3-yZn0.8CuySb2 (y = 0.075, 0.100, and 0.125). (A-C) SEM images for y = 0.075, 0.100, and 0.125; (D-G) EDS elemental mapping for the y = 0.100 sample. SEM: Scanning electron microscopy; EDS: energy-dispersive X-ray spectrometer.
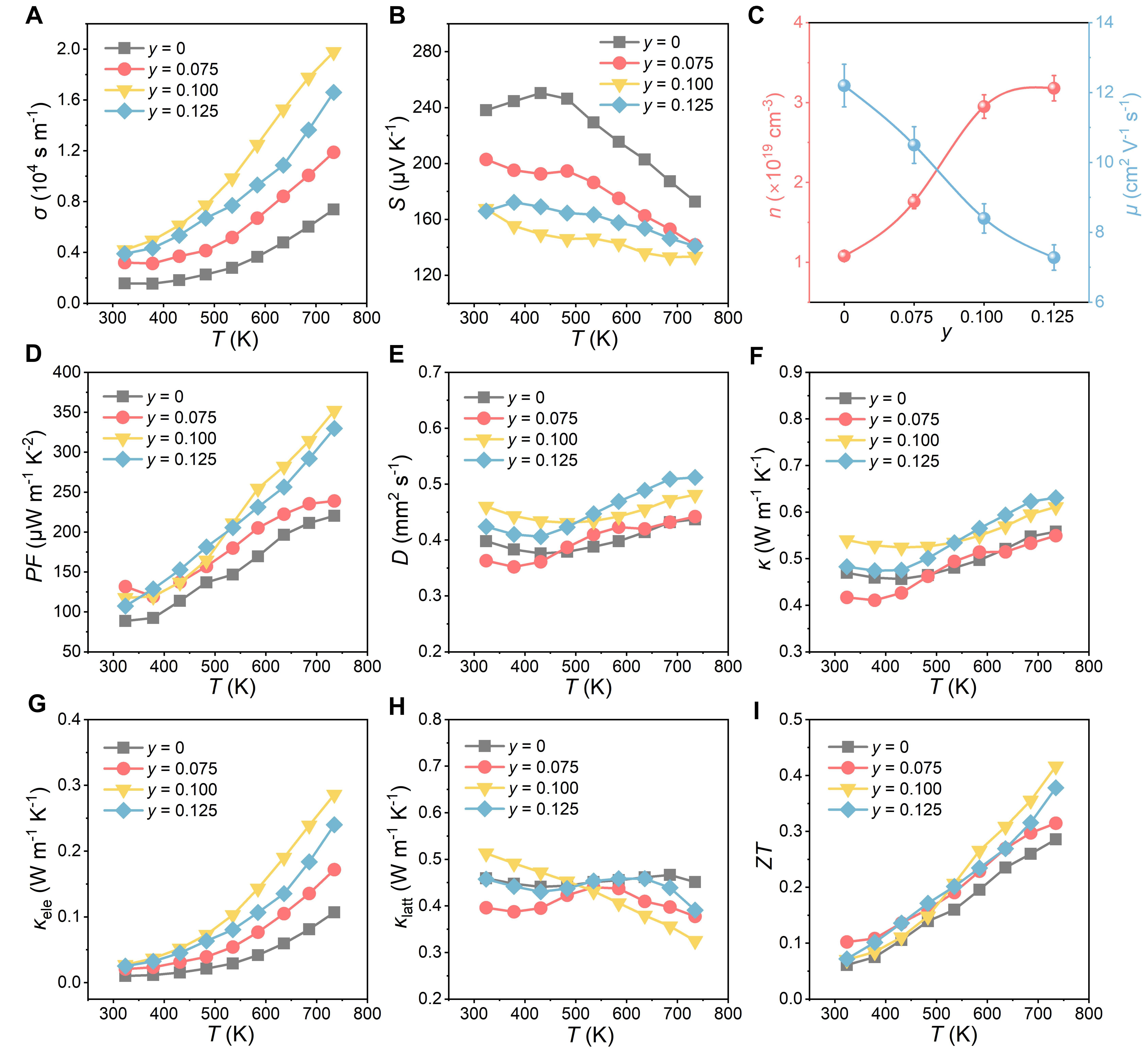
We further investigated the effects of Zn and Cu co-doping on the thermoelectric properties of the materials. Figure 7A-D shows the temperature-dependent σ, S, n, μ, and PF of Mg2.3-yZn0.8CuySb2 (y = 0, 0.075, 0.100, and 0.125). Overall, σ increases with temperature for all samples, and Cu-doped compositions exhibit significantly higher σ compared with the Cu-free sample. For example, at 735 K, the σ of the y = 0.100 sample reaches 1.98 × 104 S m-1, which is 2.68 times that of y = 0. This improvement is attributed to the band overlap caused by Zn and Cu co-doping; i.e., the top of the valence band approaches or contacts the bottom of the conduction band, such that at elevated temperatures (> 600 K), valence-band electrons are more easily thermally excited. The excitation leaves positively charged vacancies (holes) in the valence band, and the subsequent movement of electrons to fill these vacancies is equivalent to hole transport, thereby significantly increasing σ. However, S decreases markedly with Cu doping [Figure 7B]. This is because Zn and Cu co-doping pushes the EF deeper into the valence band, reducing the energy difference between the average carrier energy and EF. As a result, the heat-flow-driven energy distribution becomes narrower, leading to a lower S. Hall measurements at room temperature [Figure 7C] further confirm this trend: as y increases to 0.125, n increases while μ decreases. The rise in n arises from Cu substitution, which reduces the Eg, and Eg essentially disappears after co-doping, promoting more valence-band hole carriers. The reduction in μ is attributed to enhanced carrier-carrier scattering from higher n and increased interfacial scattering associated with partial Cu aggregation. Consistent with the Hall relation σ = neμ, n and μ vary inversely. Additionally, from the Mott relation (S
Figure 7. Thermoelectric properties of Mg2.3-yZn0.8CuySb2 (y = 0, 0.075, 0.100, and 0.125). Temperature-dependent (A) σ and (B) S; y-dependent (C) n (red) and μ (blue) at room temperature; temperature-dependent (D) PF; (E) D; (F) κ; (G) κele; (H) κlatt, and (I) ZT. PF: Power factor; ZT: dimensionless figure of merit; D: thermal diffusivity; κ: total thermal conductivity; κele: electronic thermal conductivity; κlatt: lattice thermal conductivity.
CONCLUSIONS
p-Type Mg3.1-xZnxSb2 (x = 0, 0.4, 0.6, and 0.8) and Mg2.3-yZn0.8CuySb2 (y = 0, 0.075, 0.100, and 0.125) samples were successfully synthesized by high-energy ball milling followed by hot pressing. First-principles calculations reveal that Zn and Cu co-doping at the Mg site effectively modulates the electronic band structure, driving the conduction band into direct contact with the valence band and shifting the EF deep into the valence band. This restructuring increases the hole concentration and enhances the σ. In addition, the mass fluctuation introduced by Zn and Cu substitution generates local lattice distortion, strengthens phonon scattering, and reduces the κlatt compared with the undoped material. As a result of the synergistic optimization of electrical and thermal transport, the Mg2.2Zn0.8Cu0.1Sb2 sample achieves a peak ZT of 0.42 at 735 K, representing a 147% improvement over the undoped sample. These findings demonstrate that Zn/Cu co-doping at the Mg site is an effective strategy for band-structure engineering in Mg3Sb2-based thermoelectrics. Furthermore, this approach provides a promising pathway for optimizing other Zintl-phase thermoelectric materials.
DECLARATIONS
Acknowledgments
This work was supported by Shaanxi University of Science and Technology, Queensland University of Technology (QUT), and the Xi'an International Science and Technology Cooperation Base of Semiconductor Thermoelectric Materials and Devices. The authors thank the Australian Research Council and the QUT Capacity Building Professor Program for financial support, and acknowledge the National Computational Infrastructure, supported by the Australian Government, for providing computational resources and services.
Authors’ contributions
Responsible for data collection, analysis, and interpretation: Zhang, J.; Chen, W.
Responsible for providing materials, administrative, academic, and technical support: Zhang, L.; Zhu, J.; Yang, Y.; Li, M.
Responsible for research supervision, manuscript revision, and review: Zhang, L.; Shi, X.; Chen, Z.
Availability of data and materials
The data supporting this article are included as part of the Supplementary Materials. Other raw data that support the findings of this study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was jointly supported by the Shaanxi University of Science and Technology Foundation Project (Grant No. 2017GBJ-03), the Shaanxi Province Key Laboratory of Green Preparation and Functionalization of Inorganic Materials (SKL009), the Australian Research Council, and the QUT Capacity Building Professor Program.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Jiang, B.; Yu, Y.; Cui, J.; et al. High-entropy-stabilized chalcogenides with high thermoelectric performance. Science 2021, 371, 830-4.
2. Luo, D.; Chen, H.; Chen, W.; et al. Interdependent optimization strategies for material, module, and system designs in thermoelectric devices. Device 2025, 3, 100752.
3. Luo, D.; Sun, Z.; Wang, R. Performance investigation of a thermoelectric generator system applied in automobile exhaust waste heat recovery. Energy 2022, 238, 121816.
4. Li, J. W.; Gao, H.; Han, Z.; et al. In situ engineering of grain boundary phase toward superior thermoelectric performance in Mg3(Sb,Bi)2. Adv. Mater. 2025, 37, e2503665.
5. Duparchy, A.; Naithani, H.; Ghosh, S.; et al. Poorer is better: towards robust, high performance Mg2(Si,Sn) thermoelectric material by avoiding excess Mg. Energy. Mater. 2025, 5, 500134.
6. Chen, W.; Aishwarya, K.; Sardar, K. Statistical and artificial intelligence approaches towards the optimization of thermoelectric materials synthesis: a review. Energy. Mater. 2025, 5, 500120.
7. Zhang, Z.; Ming, C.; Song, Q.; et al. Grain boundary modulation improved thermal stability of high thermoelectric performance Mg3(Sb,Bi)2-based compounds. Acta. Mater. 2025, 287, 120806.
8. Du, J.; Sun, Y.; Guo, F.; et al. Enhanced thermoelectric cooling performance of (Bi,Sb)2Te3 through platinum doping. Mater. Today. Phys. 2025, 53, 101705.
9. Yang, Q.; Yang, S.; Qiu, P.; et al. Flexible thermoelectrics based on ductile semiconductors. Science 2022, 377, 854-8.
11. He, J.; Tritt, T. M. Advances in thermoelectric materials research: looking back and moving forward. Science 2017, 357.
12. Lei, J.; Wuliji, H.; Ren, Q.; et al. Exceptional thermoelectric performance in AB2Sb2-type Zintl phases through band shaping. Energy. Environ. Sci. 2024, 17, 1416-25.
13. Ohno, S.; Imasato, K.; Anand, S.; et al. Phase boundary mapping to obtain n-type Mg3Sb2-based thermoelectrics. Joule 2018, 2, 141-54.
14. Jeong, J.; Shim, D.; Yox, P.; et al. Tuning the radius ratio to enhance thermoelectric properties in the Zintl compounds AM2Sb2 (A = Ba, Sr; M = Zn, Cd). Chem. Mater. 2023, 35, 3985-97.
15. Shuai, J.; Wang, Y.; Kim, H. S.; et al. Thermoelectric properties of Na-doped Zintl compound: Mg3-xNaxSb2. Acta. Mater. 2015, 93, 187-93.
16. Yang, J.; Zhao, S.; Liu, X.; Chen, L.; Wu, L. M. Enhancing near‐room‐temperature thermoelectric performance of n‐Type Mg3(Bi,Sb)2‐based materials through induction sintering and Mg evaporation control. Adv. Funct. Mater. 2024, 35, 2416861.
17. Ma, Y.; Shi, X.; Zhang, L.; et al. A two-step strategy improves the wide-temperature-range thermoelectric performance of Mg3+xBi1.29Sb0.7Te0.01. J. Mater. Chem. A. 2025, 13, 11406-15.
18. Shi, X.; Zhao, T.; Zhang, X.; et al. Extraordinary n-type Mg3SbBi Thermoelectrics enabled by yttrium doping. Adv. Mater. 2019, 31, e1903387.
19. Lin, Y.; Wood, M.; Imasato, K.; et al. Expression of interfacial Seebeck coefficient through grain boundary engineering with multi-layer graphene nanoplatelets. Energy. Environ. Sci. 2020, 13, 4114-21.
20. Li, J.; Jia, F.; Zhang, S.; et al. The manipulation of substitutional defects for realizing high thermoelectric performance in Mg3Sb2-based Zintl compounds. J. Mater. Chem. A. 2019, 7, 19316-23.
21. Zhang, J.; Song, L.; Iversen, B. B. Probing efficient N-type lanthanide dopants for Mg3Sb2 thermoelectrics. Adv. Sci. 2020, 7, 2002867.
22. Li, J. W.; Han, Z.; Yu, J.; et al. Wide-temperature-range thermoelectric n-type Mg3(Sb,Bi)2 with high average and peak zT values. Nat. Commun. 2023, 14, 7428.
23. Li, J.; Chetty, R.; Liu, Z.; Gao, W.; Mori, T. Enhancing The Thermoelectric Performance of n-type Mg3Sb2-based materials via Ag doping. Small 2025, 21, e2408059.
24. Priyadharshini, S.; Vijay, V.; Kamalakannan, S.; Archana, J.; Navaneethan, M. Realizing an ultralow thermal conductivity via interfacial scattering and rational-electronic band reformation in p-type Mg3Sb2. Appl. Phys. Lett. 2024, 124, 031601.
25. Cui, Y.; Zhang, X.; Duan, B.; et al. Band structure and thermoelectric properties of Al-doped Mg3-xAlxSb2 compounds. J. Mater. Sci:. Mater. Electron. 2019, 30, 15206-13.
26. Niu, Y.; Yang, C.; Zhou, T.; et al. Enhanced average thermoelectric figure of merit of p-type Zintl phase Mg2ZnSb2 via Zn vacancy tuning and hole doping. ACS. Appl. Mater. Interfaces. 2020, 12, 37330-7.
27. Xu, Z.; Shi, X.; Zhang, Y.; et al. Na/Bi-co-doping and heterogeneous interfaces leading to enhanced thermoelectric performance of p-type Mg3Sb2-based Zintls. Chem. Eng. J. 2024, 498, 155147.
28. Tiadi, M.; Trivedi, V.; Kumar, S.; et al. Enhanced thermoelectric efficiency in P-type Mg3Sb2: role of monovalent atoms codoping at Mg sites. ACS. Appl. Mater. Interfaces. 2023, 15, 20175-90.
29. Wang, H.; Chen, J.; Lu, T.; et al. Enhanced thermoelectric performance in p-type Mg3Sb2 via lithium doping. Chin. Phys. B. 2018, 27, 047212.
30. Huang, L.; Liu, T.; Mo, X.; et al. Thermoelectric performance improvement of p-type Mg3Sb2-based materials by Zn and Ag co-doping. Mater. Today. Phys. 2021, 21, 100564.
31. Fu, Y.; Zhang, X.; Liu, H.; Tian, J.; Zhang, J. Thermoelectric properties of Ag-doped compound: Mg3-xAgxSb2. J. Materiomics. 2018, 4, 75-9.
32. Bhardwaj, A.; Chauhan, N. S.; Goel, S.; et al. Tuning the carrier concentration using Zintl chemistry in Mg3Sb2, and its implications for thermoelectric figure-of-merit. Phys. Chem. Chem. Phys. 2016, 18, 6191-200.
33. Ren, Z.; Shuai, J.; Mao, J.; et al. Significantly enhanced thermoelectric properties of p-type Mg3Sb2 via co-doping of Na and Zn. Acta. Mater. 2018, 143, 265-71.
34. Agne, M. T.; Imasato, K.; Anand, S.; et al. Heat capacity of Mg3Sb2, Mg3Bi2, and their alloys at high temperature. Mater. Today. Phys. 2018, 6, 83-8.
35. Kresse, G.; Hafner, J. Ab initio molecular-dynamics simulation of the liquid-metal-amorphous-semiconductor transition in germanium. Phys. Rev. B. Condens. Matter. 1994, 49, 14251-69.
36. Kresse, G.; Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B. 1999, 59, 1758-75.
37. Li, J.; Zhang, S.; Han, K.; Sun, B.; Cao, L. Large improvement in thermoelectric performance of pressure-tuned Mg3Sb2. RSC. Adv. 2021, 12, 1149-56.
38. Li, A.; Hu, C.; He, B.; et al. Demonstration of valley anisotropy utilized to enhance the thermoelectric power factor. Nat. Commun. 2021, 12, 5408.
39. Zhang, X.; Luo, H.; Cao, X.; et al. Achieving excellent thermoelectric performance in p-type Mg3Sb2-based Zintl materials via synergistic band engineering and entropy engineering. Acta. Mater. 2025, 289, 120933.
40. Li, J. W.; Liu, W.; Xu, W.; et al. Bi-deficiency leading to high-performance in Mg3(Sb,Bi)2-based thermoelectric materials. Adv. Mater. 2023, 35, e2209119.
41. Nolas, G. S.; Morelli, D. T.; Tritt, T. M. Skutterudites: a Phonon-glass-electron crystal approach to advanced thermoelectric energy conversion applications. Annu. Rev. Mater. Sci. 1999, 29, 89-116.
42. Liu, W.; Tan, X.; Yin, K.; et al. Convergence of conduction bands as a means of enhancing thermoelectric performance of n-type Mg2Si1-xSnx solid solutions. Phys. Rev. Lett. 2012, 108, 166601.
43. Pei, Y.; Wang, H.; Snyder, G. J. Band engineering of thermoelectric materials. Adv. Mater. 2012, 24, 6125-35.
44. Tiadi, M.; Battabyal, M.; Jain, P. K.; Chauhan, A.; Satapathy, D. K.; Gopalan, R. Enhancing the thermoelectric efficiency in p-type Mg3Sb2 via Mg site co-doping. Sustain. Energy. Fuels. 2021, 5, 4104-14.
45. Xin, H. X.; Qin, X. Y.; Jia, J. H.; Song, C. J.; Zhang, K. X.; Zhang, J. Thermoelectric properties of nanocrystalline (Mg1-xZnx)3Sb2isostructural solid solutions fabricated by mechanical alloying. J. Phys. D. Appl. Phys. 2009, 42, 165403.
46. Ahmadpour, F.; Kolodiazhnyi, T.; Mozharivskyj, Y. Structural and physical properties of Mg3-xZnxSb2 (x = 0-1.34). J. Solid. State. Chem. 2007, 180, 2420-8.
47. Williamson, G.; Hall, W. X-ray line broadening from filed aluminium and wolfram. Acta. Metall. 1953, 1, 22-31.
48. Callaway, J. Model for Lattice Thermal Conductivity at Low Temperatures. Phys. Rev. 1959, 113, 1046-51.
49. Poudel, B.; Hao, Q.; Ma, Y.; et al. High-thermoelectric performance of nanostructured bismuth antimony telluride bulk alloys. Science 2008, 320, 634-8.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data



















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].