Rehmannia glutinosa nanovesicles protect cardiomyoblasts from oxidative injury
Abstract
Aim: Oxidative stress is a key driver of cardiovascular disease, underscoring the need for safe and effective antioxidant therapies. This study aims to evaluate the cardioprotective potential of plant-derived nanovesicles (PDNVs) derived from Panax ginseng (Gin) and Rehmannia glutinosa (Glu) against hydrogen peroxide (H2O2)-induced oxidative injury in cardiac cells.
Methods: PDNVs were isolated from medicinal plants via differential ultracentrifugation and characterized for morphology, diameter, stability, and cellular uptake. The antioxidant and cytoprotective effects were assessed in H2O2-injured cardiomyoblasts through cell viability, 2,2-diphenyl-1-picrylhydrazyl radical (DPPH·) radical scavenging, intracellular reactive oxygen species (ROS) and mitochondrial superoxide detection, and antioxidant enzyme (superoxide dismutase, SOD; glutathione peroxidase, GPx) activity assays. The composition of the PDNVs was determined through Coomassie Brilliant Blue staining for proteins, agarose gel electrophoresis for nucleic acids, and liquid chromatography-mass spectrometry system (LC-MS) for bioactive monomers.
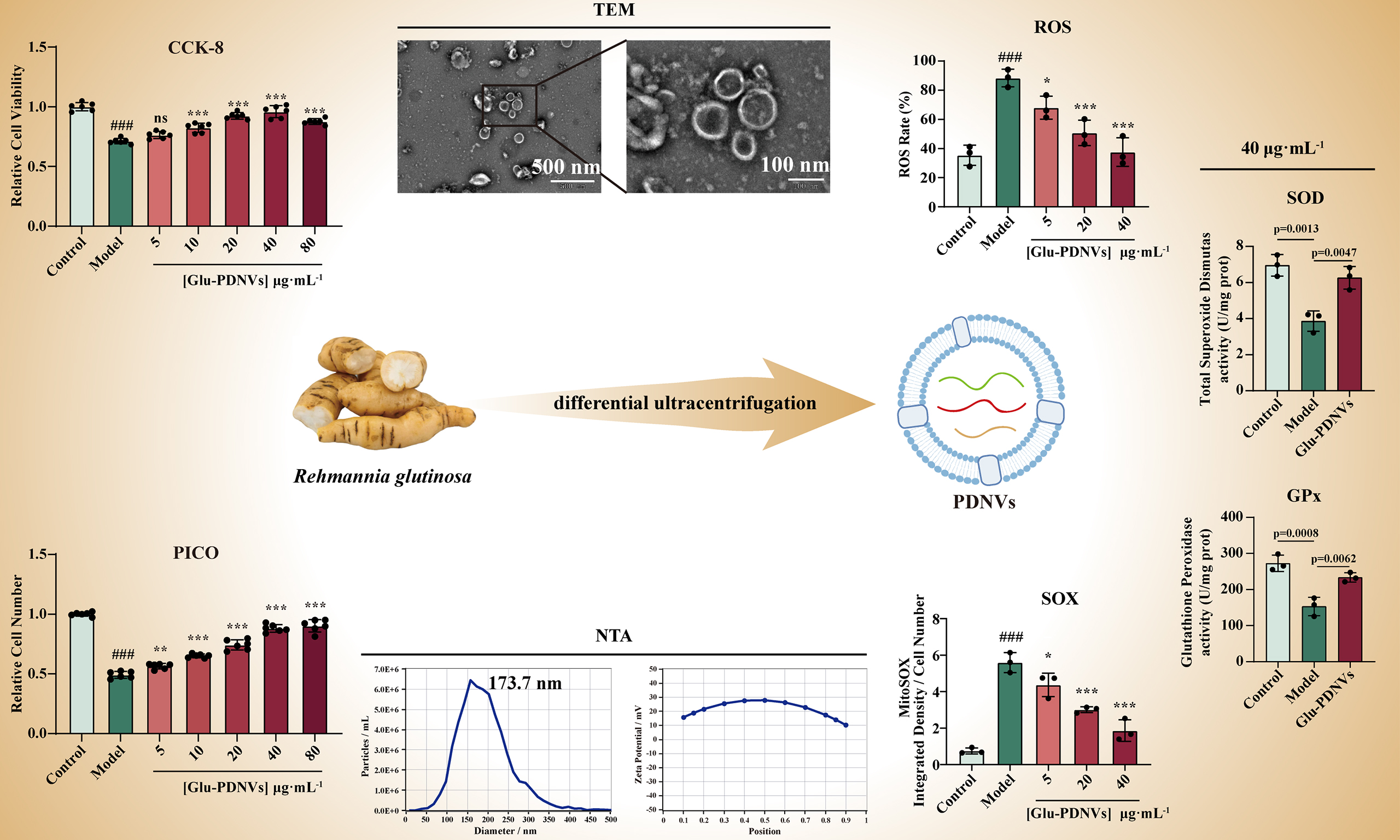
Results: Gin-PDNVs and Glu-PDNVs significantly enhanced cardiomyoblast viability under oxidative stress. Glu-PDNVs demonstrated superior efficacy at lower concentrations, with stronger ROS scavenging capacity. Compositional analysis revealed that Glu-PDNVs carry proteins, nucleic acids, and antioxidant herbal compounds such as catalpol, rehmannioside D, and acteoside. Glu-PDNVs also dose-dependently scavenged DPPH· radicals, reduced mitochondrial superoxide accumulation, and significantly restored the H2O2-induced suppression of SOD and GPx activities.
Conclusion: This study provides the first evidence that Glu-PDNVs exert potent cardioprotection by regulating ROS and superoxide homeostasis, positioning them as a promising natural nanotherapeutic platform with translational potential.
Keywords
INTRODUCTION
The extensive occurrence of redox reactions makes oxidative stress a critical factor affecting cardiac functions. Oxidative stress is defined as an imbalance where the body’s antioxidant defense system is insufficient to eliminate pro-oxidative molecules (such as reactive oxygen species, ROS), leading to oxidative modifications of key biomolecules such as proteins, lipids, and nucleic acids[1]. Such molecular damage can disrupt mitochondrial function and induce apoptosis and ferroptosis[2-4]. Ultimately, it triggers adverse cardiac remodeling in various heart diseases including myocardial infarction, cardiac ischemia-reperfusion injury, and heart failure[5,6]. Consequently, developing highly effective, biocompatible antioxidant therapeutics has become a critical focus of contemporary cardiovascular research. Cell-derived nanovesicles (CDVs) are lipid bilayer-structured particles possessing excellent biocompatibility and low immunogenicity in the delivery of bioactive molecules such as proteins and lipids from donor cells to recipient cells, thereby regulating the physiological functions of recipient cells[7,8]. Current research has focused extensively on CDVs derived from animal cells. Among these, mesenchymal stromal cell-derived extracellular vesicles (MSC-EVs) exhibit significant antioxidant properties and can regulate cellular processes such as proliferation, apoptosis, inflammation, and angiogenesis. Consequently, they show promising therapeutic potential in the treatment of various cardiovascular diseases[9-11]. However, MSC-EVs suffer from inherent limitations such as low yield and high cost, which severely hinder their clinical translation process[12-14].
Research has revealed that nanovesicles extracted from plants are similar to animal CDVs in their structure, content, and release mechanisms[15]. These are called plant-derived nanovesicles (PDNVs) and typically range from 30-200 nm[16,17]. PDNVs exhibit excellent biocompatibility, comparable to nanovesicles derived from animal cells. Moreover, studies have shown that PDNVs are efficiently internalized by recipient cells and do not induce significant cytotoxicity[18]. Importantly, PDNVs lack mammalian antigens and display lower immunogenicity[19]. In animal model experiments, PDNV administration did not cause detectable pathological damage to major organs or abnormalities in key blood biochemical parameters, indicating negligible systemic toxicity[20]. Moreover, their raw materials are abundant and do not require costly in vitro culture systems, enabling more efficient and economical large-scale production. PDNVs provide unique therapeutic advantages by preserving active herbal components and delivering superior pharmacological effects compared to the raw herbs[21-23]. Their lipid structure protects bioactive compounds from gastric and intestinal degradation, greatly enhancing oral bioavailability[24,25]. Oral administration is convenient and improves patient compliance, while other routes, such as intraperitoneal, intravenous, or intranasal delivery, and biomaterial-based cardiac patches, can further optimize biodistribution and targeting[26-30]. Given these advantages, PDNVs derived from medicinal herbs have shown promising antioxidant and cardioprotective activities. For instance, Panax ginseng (Gin)-PDNVs alleviate hydrogen peroxide (H2O2)-induced damage in Human Umbilical Vein Endothelial Cells (HUVECs) and treat cisplatin-induced cardiac injury by suppressing oxidative stress[31,32]. Similarly, Salvia miltiorrhiza-PDNVs have been demonstrated to promote angiogenesis, thereby improving myocardial ischemia-reperfusion injury[33].
Among medicinal plants with therapeutic potential, Rehmannia glutinosa (Glu) stands out due to its well-established antioxidant and cardioprotective properties. As a plant with both medicinal and food uses, Glu contains bioactive constituents, e.g., catalpol, rehmannioside D, acteoside, and polysaccharides, which have demonstrated pharmacological effects[34-37]. Catalpol, the most abundant iridoid glycoside, serves as the official quality control marker in the Chinese Pharmacopoeia, while rehmannioside D and acteoside are critical for species authentication. However, the clinical translation of these bioactive compounds faces challenges including poor stability, low solubility, and limited oral bioavailability[38,39]. Meanwhile, the application of its polysaccharides is hindered by a lack of efficient preparation methods, unclear structure-activity relationships, and insufficient in vivo evidence[40]. In contrast, PDNVs not only efficiently concentrate bioactive constituents but also feature straightforward preparation, structural stability, and significantly enhanced bioavailability.
Based on a H2O2-induced rat H9c2 cardiomyoblast injury model, this study assessed the antioxidant capacity of Glu-PDNVs in comparison with Gin-PDNVs. Furthermore, we investigated the regulatory effects of Glu-PDNVs on intracellular ROS and superoxide levels, as well as on the activities of the antioxidant enzymes superoxide dismutase (SOD) and glutathione peroxidase (GPx). This study will provide provide new support for developing herbal PDNV-based antioxidant strategies for cardiac protection.
METHODS
Cell culture and H2O2 injury model
H9c2 cells, a cardiomyoblast cell line, were kindly provided by Yang Lu and Professor Shuwen Guo at Beijing University of Chinese Medicine[41]. These cells were maintained at 37 °C with 5% CO2 and saturated humidity. The complete medium used was prepared with 4.5 g·L-1 high-glucose Dulbecco’s Modified Eagle Medium (DMEM) medium (Gibco, Grand Island, NY, USA), supplemented with 10% fetal bovine serum (FBS; Procell, Wuhan, China) and 1% penicillin-streptomycin solution (P/S; Gibco, Grand Island, NY, USA). The medium was replaced every two days, and all experiments used cells in the logarithmic growth phase. Following a 24 h adhesion period post-seeding, H9c2 cells were subjected to oxidative stress by treatment with H2O2 (MREDA, Beijing, China) in serum-free, high-glucose (4.5 g·L-1) DMEM for 4 h. Subsequently, the cells were returned to complete medium and cultured for an additional 20 h.
Isolation of PDNVs
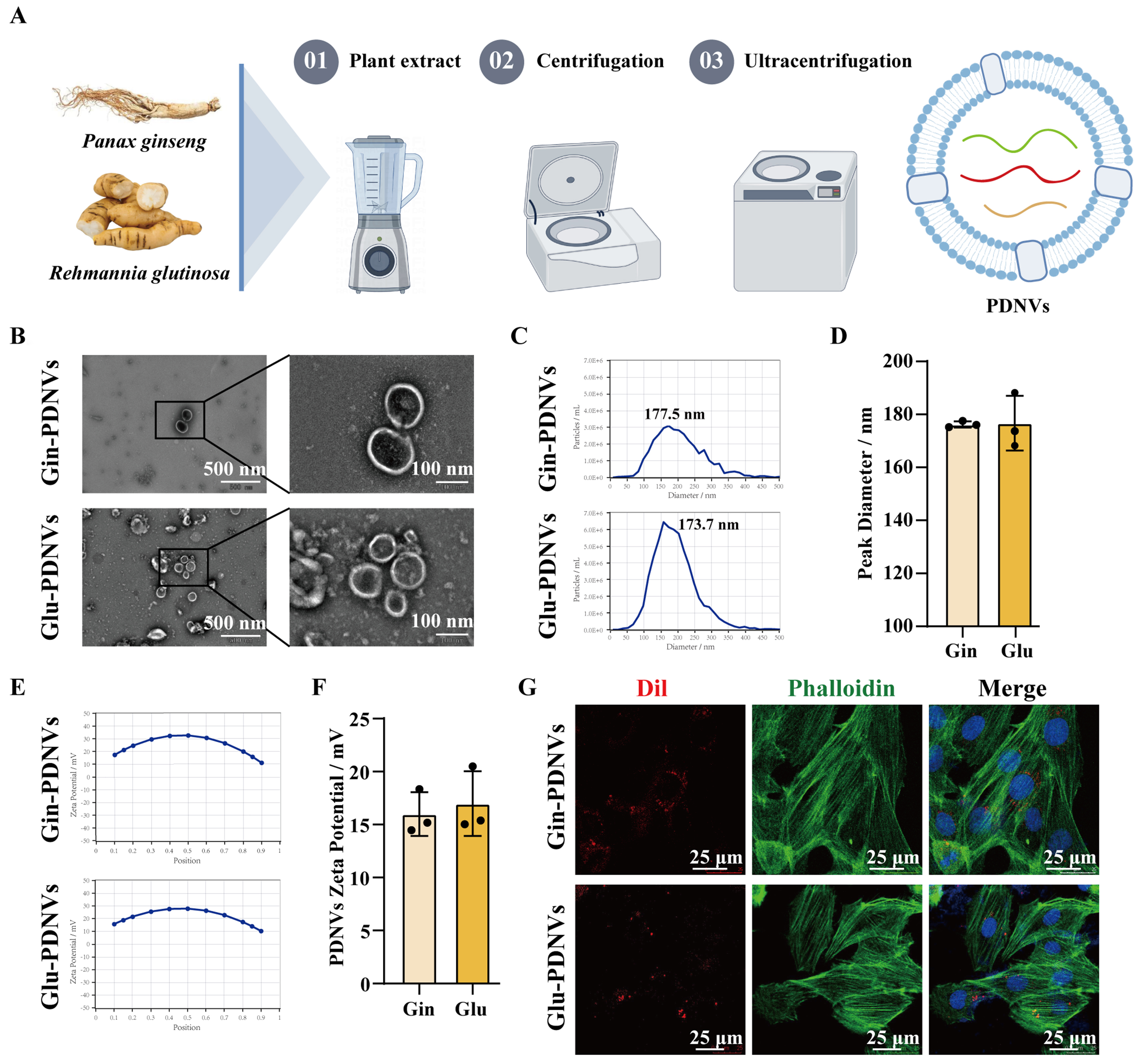
Gin and Glu were purchased from Ruifeng Beiling Chinese Herbal Medicine Co., Ltd. in Anguo City. The plant materials were authenticated by Wei Li at Beijing University of Chinese Medicine according to pharmacopeial standards. After washing, the herbs were crushed to extract juice. The resulting herbal solution was filtered through gauze, and PDNVs were subsequently isolated from the filtrate using differential ultracentrifugation (Thermo, Waltham, MA, USA) at 4 °C. Centrifugation was performed sequentially at 300 × g for 20 min, 2,000 × g for 30 min and 12,000 × g for 60 min to remove insoluble debris. The supernatant was centrifuged again at 100,000 × g for 70 min. The resulting pellet was resuspended in phosphate-buffered saline (PBS; HyClone, Logan, UT, USA) and filtered through a 0.22 µm polyethersulfone membrane. The final PDNVs were analysed for protein concentration using the BCA Protein Assay Kit (Applygen, Beijing, China). The PDNV dosage concentration used in this study corresponds to the protein concentration.
Transmission electron microscopy
A small amount of sample was pipetted onto the copper grid surface. After standing at room temperature for 10 min, residual liquid was blotted off with filter paper. Subsequently, the sample was counterstained with 3% uranyl acetate solution. The staining was performed at room temperature for 1-3 min. After allowing the sample to air dry, its morphological features were observed and analyzed using a transmission electron microscope (TEM; Hitachi, Tokyo, Japan).
Nanoparticle tracking analysis and zeta potential
The PDNV samples were appropriately diluted with PBS buffer. The particle size distribution, concentration, and zeta potential of the PDNVs were measured using a ZetaView PMX-X30 nanoparticle analyzer (Particle Metrix, Meerbusch, Germany).
Chlorophyll content assay
Chlorophyll content was measured to assess potential chloroplast contamination in PDNV preparations. Samples were mixed with an equal volume of absolute ethanol, incubated at 4 °C in the dark for 20-30 min, and then centrifuged at 12,000 rpm for 5 min. The supernatant was collected, and absorbance at 663 and 645 nm, corresponding to chlorophyll a and b, respectively, was measured using a microplate reader. Spinach homogenate and PBS served as positive and negative controls, respectively. Chlorophyll content was detected by absorbance at 663 and 645 nm.
Detection of cargo in Glu-PDNVs
To comprehensively characterize the molecular composition of PDNVs, a multi-faceted analysis was performed. Nucleic acids were extracted using TRIzol reagent (Thermo, Waltham, MA, USA), quantified via Nano Drop spectrophotometry (Implen, Munich, Germany), and assessed by Gel Red-stained agarose gel electrophoresis (Vazyme, Nanjing, China; baygene, Shanghai, China). Concurrently, PDNV proteins were separated by Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis (SDS-PAGE) and visualized with Coomassie Brilliant Blue staining (Epizyme, Shanghai, China; sparkjade, Shandong, China). For the analysis of bioactive small molecules, PDNVs were first disrupted by methanol treatment and ultrasonication. The resulting extracts, collected after centrifugation (12,000 rpm,
Cell viability assay using CCK-8
The c assay (Vazyme, Nanjing, China) was employed to assess cell viability according to the provided instructions. H9c2 cells were simultaneously treated with H2O2 and PDNVs in FBS-free DMEM for 4 h, followed by replacement with complete medium for an additional 20 h of culture. Cell Counting Kit-8 (CCK-8) reagent was then added to the cells, which were incubated at 37 °C in a 5% CO2 environment for 2 h. The absorbance at
ImageXpress pico automated cell imaging and analysis
To assess cell numbers more intuitively, Calcein-AM (Beyotime, Shanghai, China) was first diluted to the correct concentration, after which it was incubated with the cells at 37 °C and 5% CO2 for 30 min. Fluorescence images of each well were then captured using the ImageXpress Pico automated cell imaging analysis system (Molecular Devices, San Jose, CA, USA) at 10× magnification. Image segmentation and counting were then performed to generate the final statistical output of cell counts.
PDNVs uptake essay
After 30 min of incubation at 37 °C, the PDNVs were labeled with DiI dye (Beyotime, Shanghai, China). Subsequently, the stained PDNVs were co-incubated with H9c2 cells for 6 h. Following this incubation, the H9c2 cells were stained with FITC-labeled phalloidin (Solarbio, Beijing, China) according to the manufacturer’s protocol and were finally visualized under a confocal laser scanning microscope (Leica, Wetzlar, Germany).
H2O2 scavenging assay
To assess whether PDNVs directly scavenge H2O2, the residual H2O2 level was measured using the Amplex Red assay. Three experimental groups were set up: blank control (DMEM only), model group (DMEM containing 600 μmol·L-1 H2O2), and treatment group (DMEM containing 600 μmol·L-1 H2O2 and
Determination of DPPH· inhibition rate for glu-PDNVs
50 µL of the Glu-PDNVs test solution was mixed with 50 µL of the 0.200 mmol·L-1 DPPH· ethanol solution (Macklin, Shanghai, China) in a 200 µL tube. The mixture was thoroughly agitated on a vortex mixer, then left to stand in darkness at ambient temperature for 30 min, after which its absorbance at 517 nm was determined and recorded as Ai. Similarly, the absorbance (A0) was determined by mixing 50 µL of PBS with 50 µL of the DPPH· solution, and the absorbance (Aj) was determined by mixing 50 µL of the Glu-PDNVs test solution with 50 µL of anhydrous ethanol. The Scavenging rate (S) of the Glu-PDNVs against the DPPH· was determined by the following formula[42]:
Evaluation of oxidative stress
To evaluate the efficacy of PDNVs in suppressing H2O2-induced ROS accumulation in cardiomyoblasts, cells were stained using the ROS Assay Kit (Beyotime, Shanghai, China) according to the protocol, and then analysed by flow cytometry (Beckman, Brea, CA, USA).
To evaluate the level of superoxide ions within the mitochondria, cells were stained with MitoSOX and MitoTracker (Beyotime, Shanghai, China) according to the instructions provided. Finally, multi-channel images were acquired using confocal laser scanning microscopy[43].
The activities of SOD and GPx were evaluated using commercial assay kits (Beyotime, Shanghai, China) according to the manufacturer’s instructions. Total protein concentration was determined by BCA assay to normalize the activities. SOD and GPx activities were expressed as U/mg protein.
Statistical analysis
We performed one-way ANOVA with GraphPad Prism 9 (San Diego, CA, USA) to analyze the data among multiple groups. Data are presented as mean ± SD. Differences with P < 0.05 were interpreted as statistically significant. Figure preparation was supported by the following software: ImageJ, FlowJo, and Adobe Illustrator.
RESULTS
Characterization and cellular uptake of PDNVs
Ultracentrifugation was employed in this study to extract PDNVs from Gin and Glu, and these PDNVs were characterised using TEM, NTA and zeta potential analysis [Figure 1A]. TEM images revealed that Gin-PDNVs and Glu-PDNVs exhibited a typical cup-shaped morphology with intact bilayer membrane structures, resembling animal-derived EVs [Figure 1B, scale bars: 500 and 100 nm]. NTA was used to determine the particle concentration and size distribution, showing that the diameters of the two types of PDNVs were mainly distributed between 30-200 nm [Figure 1C and D]. Meanwhile, zeta potential measurements further indicated that these PDNVs possessed good stability [Figure 1E and F]. Table 1 summarizes the relevant parameters of Gin-PDNVs and Glu-PDNVs.
Figure 1. Characterization and cellular uptake of PDNVs. (A) Isolation of PDNVs from Gin and Glu by differential ultracentrifugation; (B) TEM images of Gin-PDNVs and Glu-PDNVs (Scale bars = 500 and 100 nm); NTA analysis of (C and D) diameter distribution and (E and F) zeta potential of Gin-PDNVs and Glu-PDNVs. n = 3; (G) DiI (red)-labelled PDNVs in phalloidin (green)-labelled H9c2 cells (Scale bar = 25 µm). Data are presented as mean ± SD. PDNVs: Plant-derived nanovesicles; Gin: Panax ginseng; Glu: Rehmannia glutinosa; TEM: transmission electron microscopy; NTA: nanoparticle tracking analysis; DiI: 1,1’-dioctadecyl-3,3,3’,3’-tetramethylindocarbocyanine perchlorate; SD: standard deviation; EVs: extracellular vesicles; nm: nanometer; µm: micrometer; mV: millivolt.
Corresponding parameters of PDNVs
| Parameter | Gin | Glu | ||
| Original | Normalized | Original | Normalized | |
| Plant material (g) | 50 | 50 | 250 | 50 |
| Resuspension vol. (mL) | 7.5 | 5 | 10.0 | 5 |
| Protein conc. (µg·mL-1) | 4,341 | 6,511.5 | 4,971.7 | 1,988.7 |
| Particle conc. ( 1011·mL-1) | 3.3 | 5.0 | 15 | 6.0 |
| Protein yield (µg·g-1) | 651.2 | 198.9 | ||
| Particles/protein (× 107·µg-1) | 7.6 | 30 | ||
| Particle yield (× 1010·g-1) | 4.9 | 6.0 | ||
| Size (nm) | 176.2 | 176.7 | ||
| Zeta (mV) | 16 | 17 | ||
To investigate the cellular uptake of PDNVs by H9c2 cells, DiI-labeled PDNVs were co-incubated with H9c2 cells for 6 h, after which the cytoskeleton was stained with phalloidin. Confocal microscopy revealed that DiI-labeled Gin-PDNVsand Glu-PDNVs (red spots) were all localised within the cytoskeleton [Figure 1G]. This indicates that PDNVs from these two different sources can be efficiently internalised by H9c2 cells.
Protective effects of Gin-PDNVs on H2O2-injured H9c2 cells
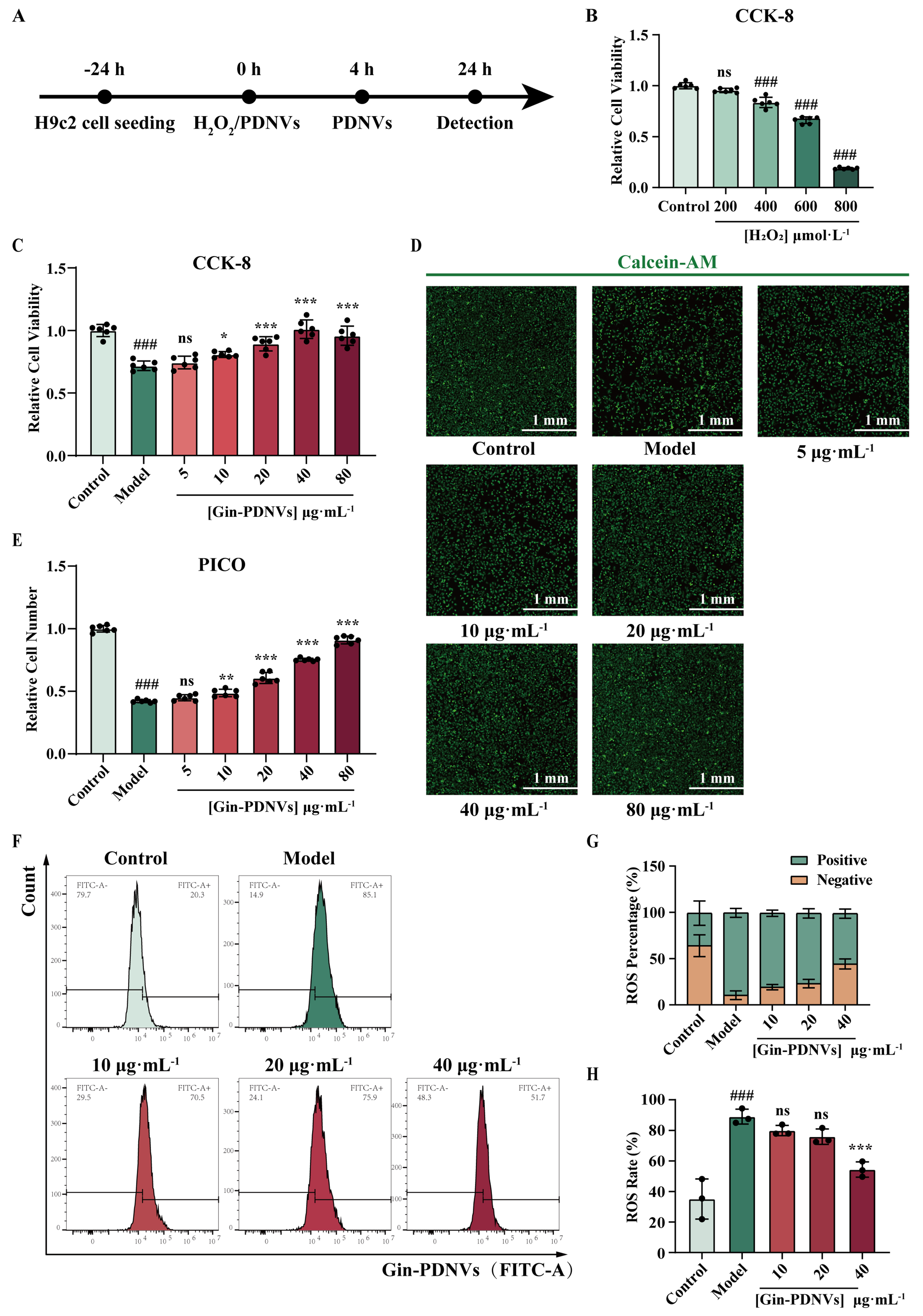
To assess the cytoprotective function of PDNVs, we evaluated the viability of H9c2 cells subjected to H2O2-induced injury [Figure 2A]. The viability of H9c2 cells exposed to H2O2 at gradient concentrations (200, 400, 600, and 800 μmol·L-1) was measured using the CCK-8 assay [Figure 2B]. Results showed that cell viability gradually decreased with increasing H2O2 concentration. At 400 μmol·L-1 H2O2, cell viability was significantly reduced compared to the control group and further declined to approximately 70% at
Figure 2. The potent cytoprotective properties of Gin-PDNVs on H9c2 cells against H2O2-induced injury. (A) Schematic illustration outlining the experimental workflow; (B) Relative viability of H9c2 cells following 4 h exposure to 200, 400, 600 and 800 µmol·L-1 H2O2. n = 6; (C) Relative viability of H9c2 cells after Gin-PDNVs treatment. n = 6; (D) Calcein-AM (green)-labeled H9c2 cells after Gin-PDNVs treatment (Scale bar = 1 mm); (E) Relative number of H9c2 cells from (D). n = 6; (F) ROS levels in H9c2 cells were measured by flow cytometry; (G) Quantitative analysis of ROS-positive and -negative cell populations. n = 3; (H) Representative flow cytometry histograms of intracellular ROS levels. n = 3. Data are presented as mean ± SD and analyzed by one-way ANOVA (###P < 0.001 vs. control; *P < 0.05, **P < 0.01, ***P < 0.001 vs. model). PDNVs: Plant-derived nanovesicles; Gin-PDNVs: Panax ginseng-derived nanovesicles; H2O2: hydrogen peroxide; CCK-8: cell counting kit-8; Calcein-AM: calcein acetoxymethyl ester; PICO: ImageXpress Pico; ROS: reactive oxygen species; FITC: fluorescein isothiocyanate; ANOVA: analysis of variance; SD: standard deviation; mm: millimeter; µmol: micromole; µg: microgram; mL: milliliter; L: liter.
In the H9c2 injury model induced by 600 μmol·L-1 H2O2, cells were treated with Gin-PDNVs at various concentrations (5, 10, 20, 40, and 80 μg·mL-1). CCK-8 assay revealed that cell viability was significantly inhibited in the model group compared to the control group [Figure 2C]. In contrast, Gin-PDNVs at
To further visually assess changes in cell number, live cells were stained with Calcein-AM and analyzed using the ImageXpress Pico automated imaging and analysis system. Representative images showed that, relative to the control, the model group had markedly fewer cells, and this reduction was gradually reversed by increasing concentrations of Gin-PDNVs [Figure 2D]. Quantitative analysis was consistent with the trends observed in the CCK-8 assay [Figure 2E].
To further validate the cytoprotective effects of Gin-PDNVs against H2O2-induced oxidative stress in H9c2 cells, we employed flow cytometry to assess ROS accumulation following treatment with Gin-PDNVs at varying concentrations (10, 20, and 40 µg·mL-1). The ROS assay showed that the model group exhibited a significant increase in ROS levels [Figure 2F-H; P < 0.001 vs. control]. In contrast, following treatment with 40 µg·mL-1 Gin-PDNVs, intracellular ROS levels were significantly reduced (P = 0.0005). These results demonstrate that Gin-PDNVs exert potent cytoprotective effects against H2O2-induced injury in H9c2 cells.
Protective effects of Glu-PDNVs on H2O2-injured H9c2 cells
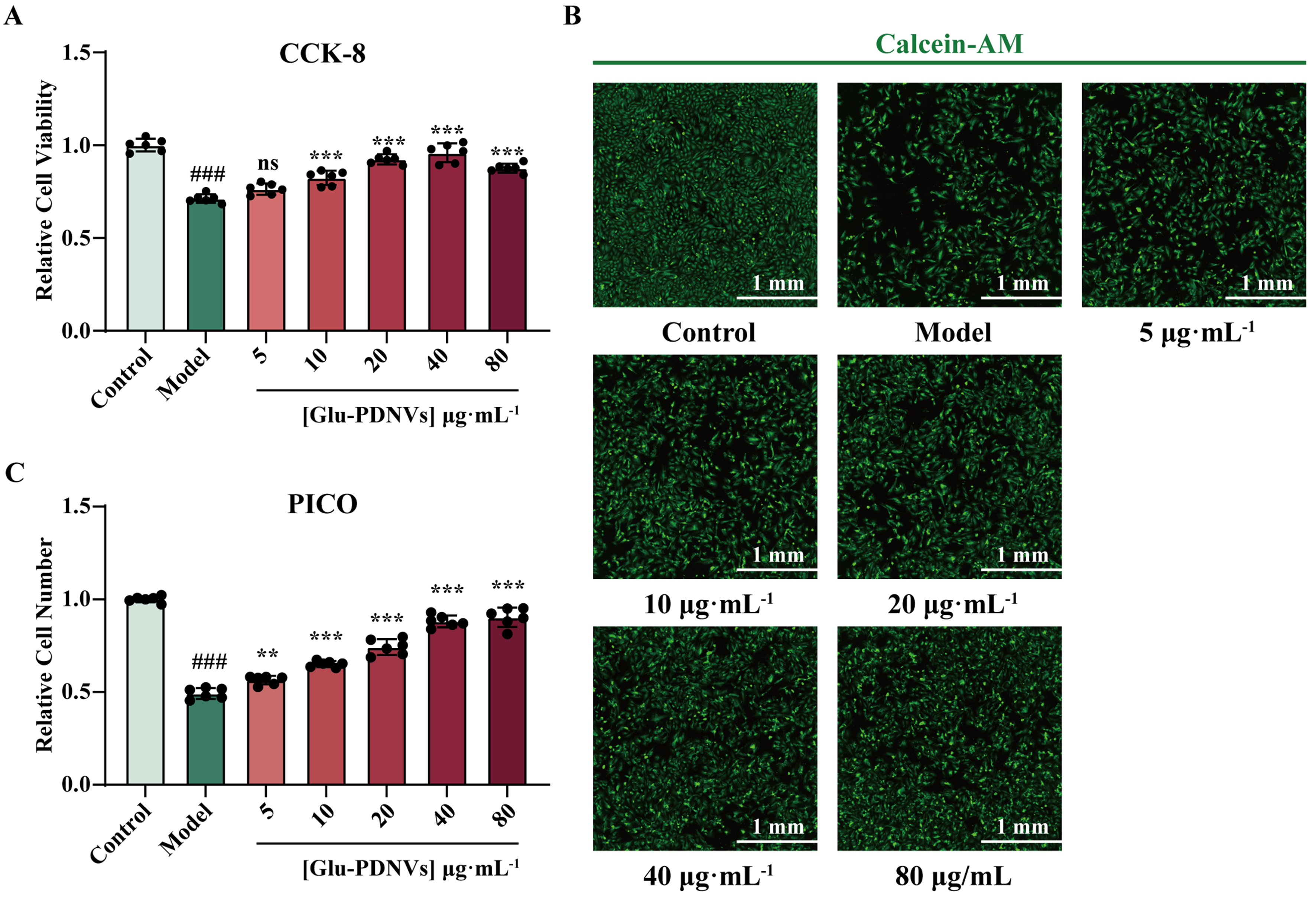
The efficacy of Glu-PDNVs was similarly evaluated. CCK-8 assay revealed that cell viability was significantly inhibited in the model group compared to the control group [Figure 3A]. In contrast, Glu-PDNVs treatment at concentrations of 10, 20, 40, and 80 µg·mL-1 resulted in significant protective effects (P < 0.001).
Figure 3. The potent cytoprotective properties of Glu-PDNVs on H9c2 cells against H2O2-induced injury. (A) Relative viability of H9c2 cells after Glu-PDNVs treatment. n = 6; (B) Calcein-AM (green)-labeled H9c2 cells after Glu-PDNVs treatment (Scale bar = 1 mm); (C) Relative number of H9c2 cells from (B). n = 6. Data are presented as mean ± SD and analyzed by one-way ANOVA (###P < 0.001 vs. control; **P < 0.01, ***P < 0.001 vs. model). PDNVs: Plant-derived nanovesicles; Glu-PDNVs: Rehmannia glutinosa-derived nanovesicles; H2O2: hydrogen peroxide; CCK-8: cell counting kit-8; Calcein-AM: calcein acetoxymethyl ester; PICO: ImageXpress Pico; ANOVA: analysis of variance; SD: standard deviation; mm: millimeter; µmol: micromole; µg: microgram; mL: milliliter; L: liter.
Automated cell imaging further confirmed these findings [Figure 3B]. The model group exhibited a clear decrease in cell number relative to the control, while Glu-PDNVs treatment led to a concentration-dependent increase in cell density. Quantitative analysis of these images yielded results consistent with the CCK-8 assay [Figure 3C].
Compositional analysis of Glu-PDNVs
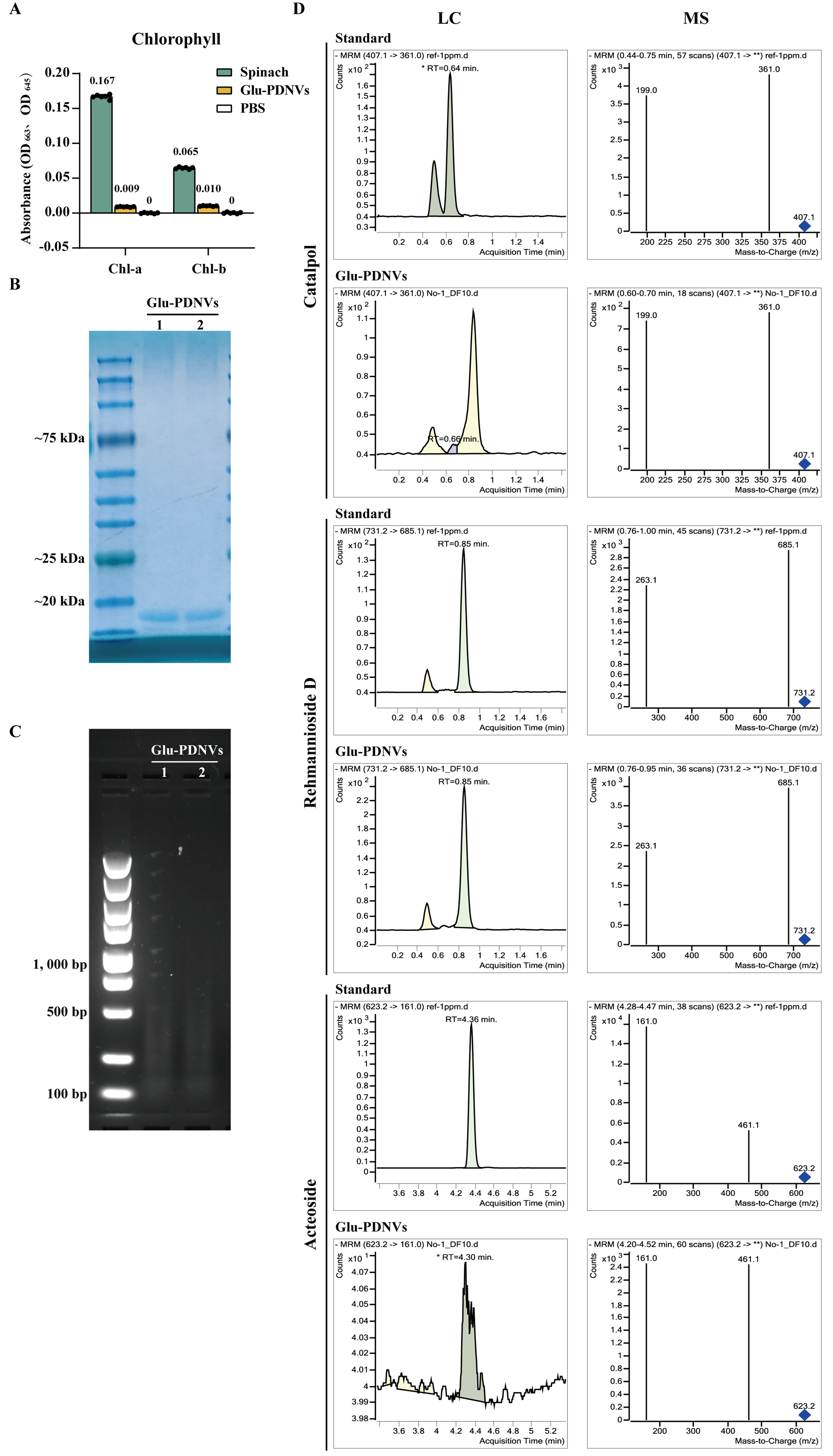
To assess chloroplast contamination, chlorophyll content was measured. PDNVs showed minimal absorbance at 663 and 645 nm (OD ≈ 0.01), over 18-fold lower than the spinach homogenate control (OD663 = 0.167), indicating effective removal of chloroplast-derived material during purification [Figure 4A]. The presence of both protein and nucleic acid cargo within Glu-PDNVs was confirmed through Coomassie Brilliant Blue staining and agarose gel electrophoresis, respectively [Figure 4B and C]. Glu-PDNVs exhibited a predominant protein band at approximately 10 kDa on SDS-PAGE and were found to contain nucleic acids at a concentration of 3,789.3 ng·mL-1 by NanoDrop spectrophotometry. To determine whether Glu-PDNVs selectively enrich the bioactive constituents of Glu, we performed LC-MS analysis targeting three representative compounds-catalpol, rehmannioside D and acteoside-all of which possess well-documented antioxidant activity[Figure 4D][34-36,44]. The absolute concentrations of these compounds and nucleic acid, as well as their values normalized to particle count and protein concentration, are presented in Table 2.
Figure 4. Compositional Analysis of Glu-PDNVs. (A) Chlorophyll content analysis of Glu-PDNV preparations. Absorbance was measured at 663 and 645 nm, the characteristic wavelengths for chlorophyll a and b, respectively. n = 3. Data are presented as mean ± SD; (B) Protein composition of Glu-PDNVs was evaluated by Coomassie Blue staining; (C) Nucleic acid composition was analyzed by agarose gel electrophoresis; (D) Representative LC-MS chromatograms for catalpol, rehmannioside D, and acteoside in Glu-PDNVs. PDNVs: Plant-derived nanovesicles; Glu-PDNVs: Rehmannia glutinosa-derived nanovesicles; LC-MS: liquid chromatography-mass spectrometry; MRM: multiple reaction monitoring; RT: retention time; m/z: mass-to-charge ratio; OD: optical density; Chl-a: chlorophyll a; Chl-b: chlorophyll b; PBS: phosphate-buffered saline; SD: standard deviation; kDa: kilodalton; bp: base pair.
Normalized nucleic acid and bioactive compound contents of Glu-PDNVs
| Nucleic acid | Catalpol | Rehmannioside D | Acteoside | |
| Original concentration (g·mL-1) | 3.79 | 0.29 | 21.91 | 0.01 |
| Per 109 particles (ng) | 0.25 | 0.02 | 1.46 | 8.67 10-4 |
| Per μg protein (ng) | 0.76 | 0.06 | 4.41 | 2.61 10-3 |
| Concentration at 40 ug/mL protein (ug·mL-1) | 0.03 | 2.3310-3 | 0.18 | 1.05 10-4 |
Glu-PDNVs protect H9c2 cells against H2O2-induced oxidative injury
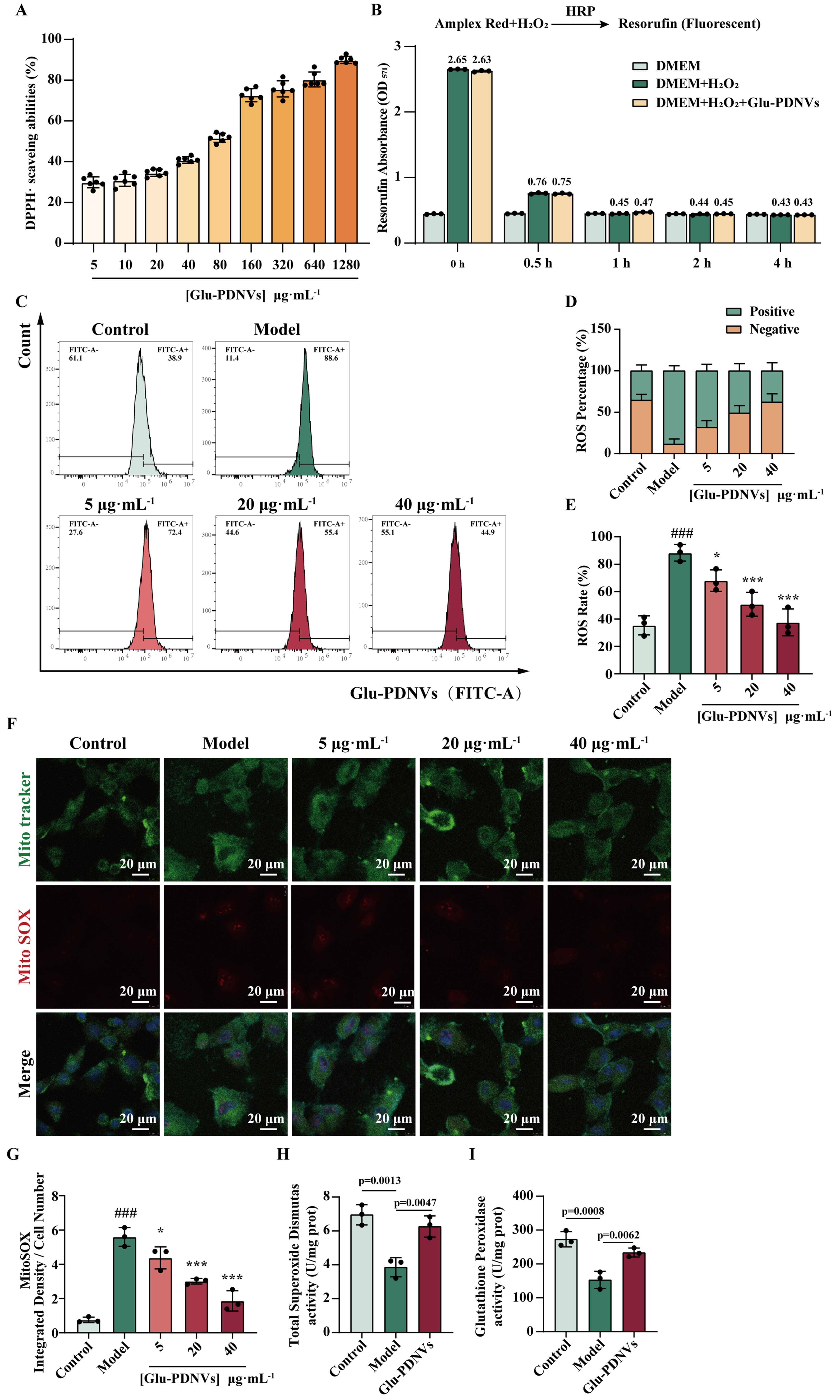
Measurement of the DPPH· inhibition rate indicates that PDNVs exhibit significant antioxidant activity, which increases with concentration [Figure 5A]. To exclude the possibility that PDNVs directly quench H2O2, we measured H2O2 consumption using the Amplex Red fluorescence assay. Results showed that the difference in OD values between the PDNV-treated group and the H2O2-only group remained minimal
Figure 5. Glu-PDNVs effectively protect H9c2 cells from oxidative damage caused by H2O2. (A) The inhibition rate of Glu-PDNVs against DPPH· radicals at gradient concentrations. n = 6; (B) Direct H2O2 scavenging capacity of Glu-PDNVs. n = 3; (C) ROS levels in H9c2 cells were quantified by flow cytometry; (D) Quantitative analysis of ROS-positive and -negative cell populations; (E) Representative flow cytometry histograms of intracellular ROS levels. n = 3; (F) Representative fluorescence images and (G) statistical analysis of MitoTracker and MitoSOX staining in H9c2 cells. n = 3; Effects of Glu-PDNVs on endogenous antioxidant enzyme activities in H2O2-injured H9c2 cells, showing (H) SOD activity and (I) GPx activity. n = 3. Data are presented as mean ± SD and analyzed by one-way ANOVA (###P < 0.001 vs. control; *P < 0.05, ***P < 0.001 vs. model). PDNVs: Plant-derived nanovesicles; Glu-PDNVs: Rehmannia glutinosa-derived nanovesicles; H2O2: hydrogen peroxide; HRP: horseradish peroxidase; Amplex Red: 10-acetyl-3,7-dihydroxyphenoxazine; DMEM: Dulbecco’s Modified Eagle Medium; DPPH·: 2,2-diphenyl-1-picrylhydrazyl radical; ROS: reactive oxygen species; FITC: fluorescein isothiocyanate; MitoTracker: mitochondrial tracker dye; MitoSOX: mitochondrial superoxide indicator; SOD: superoxide dismutase; GPx: glutathione peroxidase; OD: optical density; ANOVA: analysis of variance; SD: standard deviation; µg: microgram; mL: milliliter; µm: micrometer.
Flow cytometry analysis of intracellular ROS levels showed a significant increase in the model group [Figure 5C-E; P < 0.001 vs. control]. In contrast, Glu-PDNVs reduced ROS levels in a concentration-dependent manner, with significant inhibition observed at 5 (P = 0.0339), 20 (P = 0.0006), and 40 µg·mL-1 (P < 0.0001). Furthermore, compared to Gin-PDNVs, Glu-PDNVs exhibited a more pronounced inhibitory effect on intracellular ROS.
Mitochondrial superoxide (MitoSOX), the primary superoxide anion serves as a key molecular marker of oxidative stress. Confocal analysis demonstrated that MitoSOX fluorescence intensity was markedly higher in the model group than in controls under identical cell number conditions. This elevation was dose-dependently suppressed by Glu-PDNVs, reaching significance at concentrations of 20 and 40 µg·mL-1 [Figure 5F and G].
To further investigate the mechanisms underlying the cytoprotective effects of Glu-PDNVs, we examined the activities of key antioxidant enzymes SOD and GPx. Results showed that H2O2 treatment significantly reduced SOD activity in H9c2 cells (P < 0.01 vs. control). whereas 40 μg·mL-1 Glu-PDNVs significantly restored SOD activity [Figure 5H]. Similarly, GPx activity was markedly suppressed by H2O2 exposure and was significantly elevated upon Glu-PDNVs treatment [Figure 5I]. These results demonstrate that Glu-PDNVs enhance the cellular antioxidant defense capacity not only through direct radical scavenging but also by upregulating the activity of endogenous antioxidant enzymes.
DISCUSSION
The present study demonstrates, for the first time, that Glu-PDNVs possess significant antioxidant and cardioprotective effects against H2O2-induced oxidative injury in H9c2 cardiomyoblasts. Comparative analysis revealed that Glu-PDNVs exhibited protective effects slightly superior to those of Gin-PDNVs under equivalent experimental conditions. Notably, NTA showed that at the same protein concentration
Beyond their bioactivity, Glu-PDNVs offer practical advantages for translational development. Glu has a short growth cycle with well-established cultivation techniques and high biomass yield, enabling cost-effective and scalable production. In contrast, Gin requires 4-6 years of growth before harvest, which limits its economic feasibility for large-scale vesicle production. The combination of potent antioxidant activity and superior production efficiency positions Glu-PDNVs as a promising and sustainable nanotherapeutic platform for myocardial protection.
Unlike animal CDVs, PDNVs not only contain fundamental components such as lipids, proteins, and nucleic acids, but are also rich in various pharmacologically active natural phytochemicals. Gin-PDNVs exhibit antioxidant activity with their known content of multiple ginsenosides, e.g. Rg1, Re, Rg3, and Rb1, all of which were reported to possess antioxidant activity[45,46]. In this study, LC-MS analysis identified catalpol, rehmannioside D and acteoside in Glu-PDNVs. Rehmannioside D and catalpol both belong to the iridoid glycosides class. Previous studies have demonstrated that both compounds exhibit potent antioxidant activity in the PC-12 cell model, and catalpol has been shown to mitigate oxidative stress in myocardial ischemia-reperfusion injury[35,47]. Rehmannioside D shares structural and functional similarities with catalpol, suggesting it may also exert antioxidative stress effects in myocardial tissue. Furthermore, its high enrichment within Glu-PDNVs indicates that rehmannioside D could be a key component mediating the effects of Glu-PDNVs, though its specific pharmacological efficacy requires further validation. While these findings highlight rehmannioside D as a potential key contributor, it is important to emphasize that the cardioprotective effect of Glu-PDNVs is unlikely to result from a single compound alone. We identify catalpol, rehmannioside D, and acteoside as quantifiable, characteristic chemical tracers, demonstrating that PDNVs naturally encapsulate bioactive constituents from their parent plant. However, PDNVs also contain other bioactive molecules, including peptides, lipids, and additional small-molecule compounds, which could synergistically contribute to the overall therapeutic effect.
Despite these promising findings, several limitations of the present study should be acknowledged. First, while PDNVs function as integrated nanotherapeutic systems with synergistic multi-component interactions, the relative contribution of individual compounds to the observed effects remains unclear. Future studies testing the biological activity of purified compounds at physiologically relevant levels, both in vitro and
In conclusion, this study provides the first evidence that Glu-PDNVs possess significant antioxidant and cardioprotective effects against oxidative stress in H9c2 cardiomyoblasts. Mechanistically, Glu-PDNVs attenuated cellular damage by reducing intracellular ROS and MitoSOX levels, while restoring the activities of key antioxidant enzymes, SOD and GPx. Their superior efficacy compared to Gin-PDNVs under equivalent conditions may be attributed to a higher vesicle yield per unit protein and the enrichment of bioactive compounds, particularly rehmannioside D. While these findings establish Glu-PDNVs as a promising and sustainable nanotherapeutic platform derived from Glu, further investigations using in vivo models and human-derived cardiomyocytes are necessary to validate their translational potential for myocardial protection.
DECLARATIONS
Acknowledgments
We thank Yang Lu and Professor Shuwen Guo at Beijing University of Chinese Medicine for providing the H9c2 cells for this study. We also acknowledge Wei Li for authenticating the plant materials. Some of the graphic elements in the figures were sourced from the Figdraw material library.
Authors’ contributions
Conducted experiments with subsequent data analysis, and wrote and revised the manuscript: Fan H
Participated in the experiment of mitochondrial superoxide detection: Zhao S
Participated in the experiment of cell culture: Di Y
Participated in the experiment of Isolation of PDNVs, reviewed and edited the manuscript: Yu Y
Participated in the experiment of agarose gel electrophoresis, reviewed and edited the manuscript: Sun Y
Designed the experiment, supervised the project, reviewed and edited the manuscript: Wang J, Li C, Wang Y
All authors have read and approved the final version of the article.
Availability of data and materials
All relevant data are included within the paper. The data that support the findings of this study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool DeepL Write and DeepSeek (version V3.2, released 2025-12-01) were used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
This work was supported by National Natural Science Foundation of China (82505382).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Xu X, Pang Y, Fan X. Mitochondria in oxidative stress, inflammation and aging: from mechanisms to therapeutic advances. Signal Transduct Target Ther 2025;10:190.
2. Peoples JN, Saraf A, Ghazal N, Pham TT, Kwong JQ. Mitochondrial dysfunction and oxidative stress in heart disease. Exp Mol Med 2019;51:1-13.
3. Zhang Z, Yang J, Zhou Q, et al. The role and mechanism of the cGAS-STING pathway-mediated ROS in apoptosis and ferroptosis induced by manganese exposure. Redox Biol 2025;85:103761.
4. Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol 2021;22:266-82.
5. Cinato M, Andersson L, Miljanovic A, et al. Role of perilipins in oxidative stress-implications for cardiovascular disease. Antioxidants 2024;13:209.
6. Guo Y, Zhang W, Zhou X, et al. Roles of ferroptosis in cardiovascular diseases. Front Cardiovasc Med 2022;9:911564.
7. Welsh JA, Goberdhan DCI, O’Driscoll L, et al.; MISEV Consortium. Minimal information for studies of extracellular vesicles (MISEV2023): from basic to advanced approaches. J Extracell Vesicles. 2024;13:e12404.
8. Wang X, Xia J, Yang L, Dai J, He L. Recent progress in exosome research: isolation, characterization and clinical applications. Cancer Gene Ther 2023;30:1051-65.
9. Xia C, Dai Z, Jin Y, Chen P. Emerging antioxidant paradigm of mesenchymal stem cell-derived exosome therapy. Front Endocrinol 2021;12:727272.
10. Di Bella MA. Overview and update on extracellular vesicles: considerations on exosomes and their application in modern medicine. Biology 2022;11:804.
11. Pan Y, Wu W, Jiang X, Liu Y. Mesenchymal stem cell-derived exosomes in cardiovascular and cerebrovascular diseases: From mechanisms to therapy. Biomed Pharmacother 2023;163:114817.
12. Di Y, Zhao S, Fan H, et al. Mass production of rg1-loaded small extracellular vesicles using a 3D bioreactor system for enhanced cardioprotective efficacy of doxorubicin-induced cardiotoxicity. Pharmaceutics 2024;16:593.
13. Song Y, Wang B, Zhu X, et al. Human umbilical cord blood-derived MSCs exosome attenuate myocardial injury by inhibiting ferroptosis in acute myocardial infarction mice. Cell Biol Toxicol 2021;37:51-64.
14. Gong ZT, Xiong YY, Ning Y, et al. Nicorandil-pretreated mesenchymal stem cell-derived exosomes facilitate cardiac repair after myocardial infarction via promoting macrophage M2 polarization by targeting miR-125a-5p/TRAF6/IRF5 signaling pathway. Int J Nanomedicine 2024;19:2005-24.
15. Zhao B, Lin H, Jiang X, et al. Exosome-like nanoparticles derived from fruits, vegetables, and herbs: innovative strategies of therapeutic and drug delivery. Theranostics 2024;14:4598-621.
16. Yang J, Wang Y, Fu Y, et al. Plant extracellular vesicles: a promising bionic medicine platform for disease treatment and drug delivery. Interdiscip Med 2025;3:e20240101.
17. Pinedo M, de la Canal L, de Marcos Lousa C. A call for Rigor and standardization in plant extracellular vesicle research. J Extracell Vesicles 2021;10:e12048.
18. Li S, Ma S, Wang P, et al. Flos Puerariae-derived exosomes-like nanoparticles ameliorate alcoholic liver disease by targeting IL-11-related signaling pathways and reactive oxygen species. Chem Eng J 2025;512:161896.
19. Dad HA, Gu TW, Zhu AQ, Huang LQ, Peng LH. Plant exosome-like nanovesicles: emerging therapeutics and drug delivery nanoplatforms. Mol Ther 2021;29:13-31.
20. Xie J, Lan H, Ma H, et al. Oral Epimedium nanovesicles promote hematopoietic stem cell regeneration via gut-microbiota-bone marrow axis to mitigate chemotherapy-induced myelosuppression. Nano Res 2026;19:94908148.
21. Yang Y, Yang L, Deng H, et al. Coptis chinensis-derived extracellular vesicle-like nanoparticles delivered miRNA-5106 suppresses NETs by restoring zinc homeostasis to alleviate colitis. J Nanobiotechnology 2025;23:444.
22. Wang Y, Zhou Y, Wu Q, et al. Honeysuckle-derived nanovesicles regulate gut microbiota for the treatment of inflammatory bowel disease. Adv Sci 2025;12:e05208.
23. Fu J, Liu Z, Feng Z, et al. Platycodon grandiflorum exosome-like nanoparticles: the material basis of fresh platycodon grandiflorum optimality and its mechanism in regulating acute lung injury. J Nanobiotechnology 2025;23:270.
24. Han X, Zheng W, Sun Z, et al. Plant-derived exosomes: unveiling the similarities and disparities between conventional extract and innovative form. Phytomedicine 2025;145:157087.
25. Fang Z, Liu K. Plant-derived extracellular vesicles as oral drug delivery carriers. J Control Release 2022;350:389-400.
26. Zhang M, Xu X, Su L, et al. Oral administration of Sophora Flavescens-derived exosomes-like nanovesicles carrying CX5461 ameliorates DSS-induced colitis in mice. J Nanobiotechnology 2024;22:607.
27. Ou X, Wang H, Tie H, et al. Novel plant-derived exosome-like nanovesicles from Catharanthus roseus: preparation, characterization, and immunostimulatory effect via TNF-α/NF-κB/PU.1 axis. J Nanobiotechnology 2023;21:160.
28. Sui Y, Sun X, Liu Q, et al. Mori fructus-derived extracellular vesicle-like nanoparticles regulate dyslipidemia and prevent atherosclerosis progression via miRNAs. Phytomedicine 2025;146:157128.
29. Xu Y, Zhao JY, Xu XY, Liu YD, Li YW, Peng LH. Engineered biomimetic nanorobots orchestrate targeted nose-to-brain delivery to resolve neuron-glia entanglement against Parkinson’s disease. Small 2026;22:e13394.
30. Zhou HH, Zhou X, Pei J, et al. A fibrin gel-loaded Gouqi-derived nanovesicle (GqDNV) repairs the heart after myocardial infarction by inhibiting p38 MAPK/NF-κB p65 pathway. J Nanobiotechnology 2025;23:535.
31. Fan L, Jia X, Dong F, et al. Ginseng-derived nanoparticles accelerate diabetic wound healing by modulating macrophage polarization and restoring endothelial cell function. Mater Today Bio 2025;34:102143.
32. Yang S, Guo J, Chen D, et al. The cardioprotective effect of ginseng derived exosomes via inhibition of oxidative stress and apoptosis. ACS Appl Bio Mater 2025;8:814-24.
33. Zhang S, Xia J, Zhu Y, Dong M, Wang J. Establishing Salvia miltiorrhiza-derived exosome-like nanoparticles and elucidating their role in angiogenesis. Molecules 2024;29:1599.
34. Huang C, Cui Y, Ji L, et al. Catalpol decreases peroxynitrite formation and consequently exerts cardioprotective effects against ischemia/reperfusion insult. Pharm Biol 2013;51:463-73.
35. Zhang L, Lu RR, Wang HH, Li M, Feng WS, Zheng XK. Protective effect and mechanism of rehmannioside D on PC-12 cells injury induced by corticosterone. Chin Tradit Herbal Drugs 2022;53:3385-93. (in Chinese).
36. Zhu X, Sun M, Guo H, et al. Verbascoside protects from LPS-induced septic cardiomyopathy via alleviating cardiac inflammation, oxidative stress and regulating mitochondrial dynamics. Ecotoxicol Environ Saf 2022;233:113327.
37. Peng H. Protection of rehmannia glutinosa polysaccha-rides on rats with myocardial ischemia-reperfusion injury. J Liaoning Univ Tradit Chin Med 2016;18:39-42.
38. Wang M, Zheng X, Yang M, et al. Iridoid for drug discovery: structural modifications and bioactivity studies. Med Chem Res 2024;33:2329-46.
39. Zhang FK, Jia KX, Wang H, et al. Acteoside as a rising star for clinical treatment: Current fundamental research and future outlooks. J Integr Med 2026;24:7-23.
40. Ren H, Zhang S, Qi C, et al. Research progress on extraction technology and biological activity and mechanism of polysaccharides from Rehmannia glutinosa. J Food Saf Qual 2023;14:222-31. Available from: https://d.wanfangdata.com.cn/periodical/spaqzljcjs202304028. [Last accessed on 20 Apr 2026].
41. Li Y, Du T, Zhang Y, et al. Cardioprotective effect of Yiqi Huoxue decoction on post-myocardial infarction injury mediated by Ca2+ flux through MAMs. Front Cardiovasc Med 2025;12:1596757.
42. Wang X, Zou S, Li H, Deng Z, Pan Y, Li H. Predictive modeling of antioxidant synergies in fruits and vegetables: a multi-objective optimization approach for functional food design. Food Chem 2025;493:146005.
43. Zhao S, Fan H, Yang S, et al. Artificial cell derived vesicles from Ginsenoside Rg1-primed mesenchymal stromal cells mitigate oxidative stress and DNA damage in myocardial ischemic/reperfusion injury. Nano Research 2025;18:94907535.
44. Xiao Y, Ren Q, Wu L. The pharmacokinetic property and pharmacological activity of acteoside: a review. Biomed Pharmacother 2022;153:113296.
45. Kim J, Zhu Y, Chen S, et al. Anti-glioma effect of ginseng-derived exosomes-like nanoparticles by active blood-brain-barrier penetration and tumor microenvironment modulation. J Nanobiotechnology 2023;21:253.
46. Paik S, Song GY, Jo EK. Ginsenosides for therapeutically targeting inflammation through modulation of oxidative stress. Int Immunopharmacol 2023;121:110461.
47. Li DQ, Bao YM, Zhao JJ, Liu CP, Liu Y, An LJ. Neuroprotective properties of catalpol in transient global cerebral ischemia in gerbils: dose-response, therapeutic time-window and long-term efficacy. Brain Res 2004;1029:179-85.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data



















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].