Stem cell therapy for rare neurological diseases: translational research opportunities
Abstract
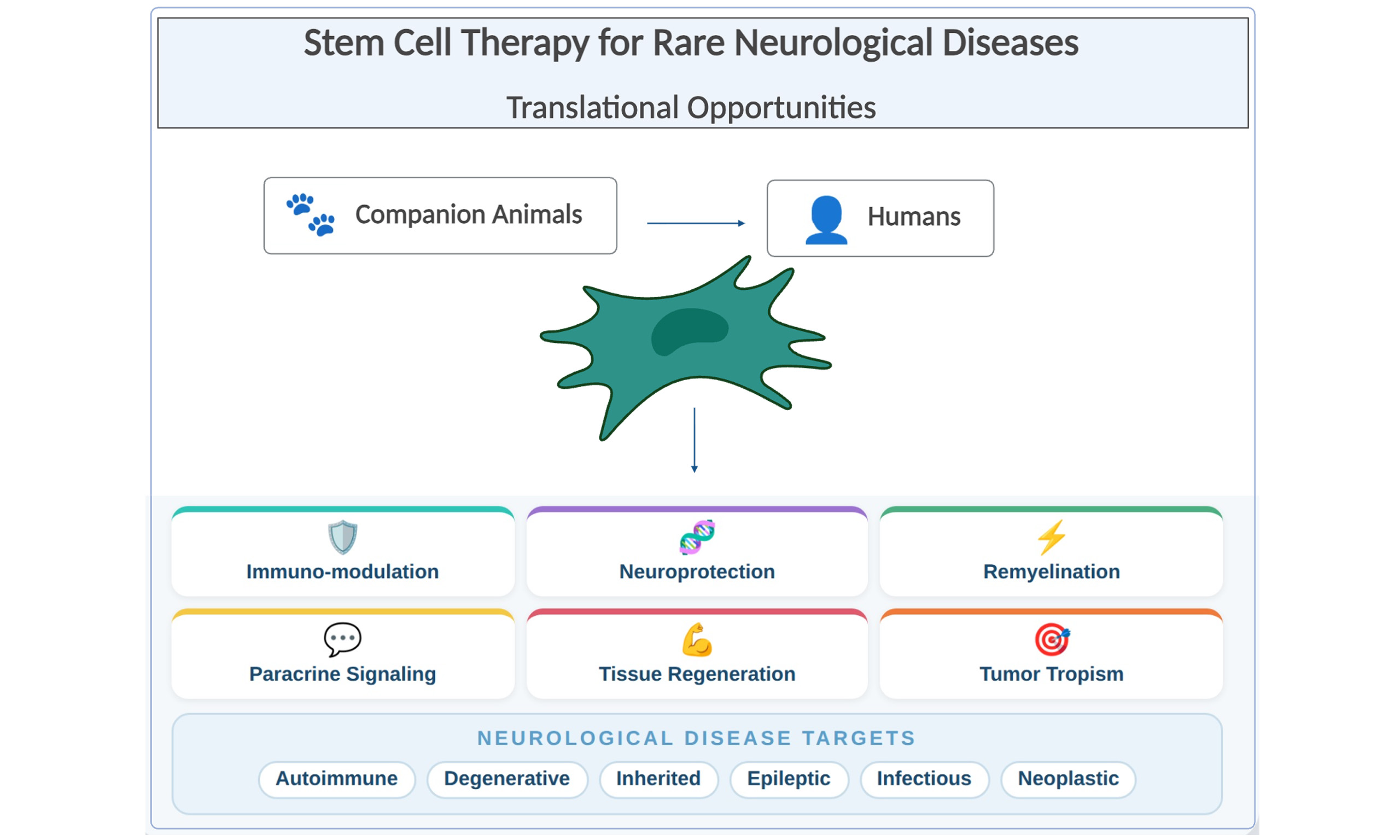
Stem cell therapy is being explored for several rare neurological conditions in humans given their potential immunomodulatory, reparative, and regenerative capabilities. Existing standard treatments for most neuroinflammatory and neurodegenerative disorders are primarily palliative, focusing on symptom management rather than addressing underlying disease pathology. The genetic and pathophysiological parallels between many human and animal neurological diseases suggest that companion animals may serve as valuable translational models to drive stem cell research forward. Several rare human neurological conditions with companion animal (particularly canine) correlates include fulminant multiple sclerosis (MS), myasthenia gravis, amyotrophic lateral sclerosis (ALS), Duchenne Muscular Dystrophy (DMD), Dravet and Lennox-Gastaut Syndromes, Globoid Cell Leukodystrophy/Krabbe Disease, viral encephalitis, and glioblastoma multiforme (GBM). Validating the safety and feasibility of stem cell transplantation in companion animal models has enabled the development and expansion of innovative therapies. Stem cell therapy may hold promise as a novel treatment option for some rare and aggressive human and companion animal neurological diseases with limited treatment options.
Keywords
INTRODUCTION
Rare neurological diseases remain particularly difficult to treat in humans and animals, with few long-term effective therapies despite decades of research, due in part to low patient numbers and patient heterogeneity[1-3]. Across species, neurological diseases are often progressive, debilitating, and resistant to conventional therapies. This is likely related to shared mechanisms of neuroinflammation and neurodegeneration, including blood-brain barrier disruption, microglial activation, oxidative stress, and cytokine release[4,5].
Although rodent models are commonly used in drug development, several naturally occurring neurological diseases in dogs, and some conditions in cats, more closely resemble human disorders, making companion animals better suited for translational research[6]. Some species differences including the low likelihood of forming atherosclerotic lesions in companion animals limit the translational potential for certain conditions[7,8]; however, many underlying inflammatory and degenerative processes are similar. Spontaneous genetic disorders in pets have already enabled gene discovery and therapeutic development for both animals and humans[9]. These success stories demonstrate how veterinary clinical trials can bridge the gap between preclinical rodent studies and human trials, reducing failure rates and accelerating therapy development for rare or orphan diseases with limited treatment options[10].
Many rare neurological conditions fail multimodal therapy, and patients are left with persistent or progressive debilitation, long-term medication-associated negative side effects, and exhaustive financial burden[3]. Standard therapies primarily target symptom management, with few options to address the underlying cause of disease[2]. Stem cell therapy has emerged as a promising option for a variety of inflammatory and degenerative conditions to target disease at the source[11]. Dogs have been an essential large-animal model for the development of stem-cell-based therapies because they naturally share key biological, genetic, and disease characteristics with humans[12]. Spontaneously occurring neurological conditions, hematologic disorders, neoplastic processes, genetic disorders, and immune deficiencies in dogs closely mirror human diseases. These models enable more accurate evaluation of immunomodulation, immune tolerance, and stem cell engraftment and transplantation biology in clinically relevant settings. Canine studies were pivotal in establishing donor matching techniques, conditioning regimens, graft collection methods, and graft-versus-host disease prevention strategies that directly enabled modern human stem cell therapy protocols[12-14]. These models also demonstrated the feasibility of using stem-cell-induced immune tolerance to support solid-organ transplantation without lifelong immunosuppression, a concept now being translated into human medicine[12] Collectively, the canine model has provided indispensable translational insights that accelerated the safe and effective adoption of stem cell therapies for human patients.
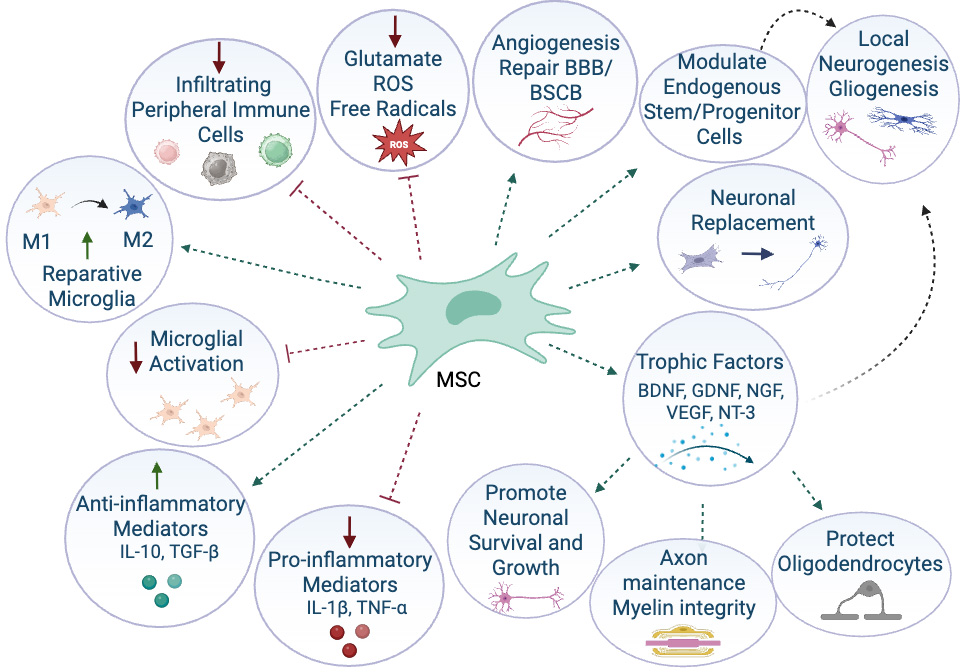
Mesenchymal stromal/stem cell (MSC) therapy has emerged as a promising approach for treating a wide range of non-hematologic inflammatory and degenerative diseases in humans and animals, owing to their potent immunomodulatory and anti-inflammatory properties. These properties enable MSCs to regulate immune responses, promote tissue repair, and modulate disease processes at the molecular level [Figure 1][15-19]. Their ability to home to sites of inflammation within the central nervous system and to exert direct and paracrine effects on the local immune system makes MSCs attractive therapeutic candidates for a variety of neurological conditions. Their low level of major histocompatibility (MHC) class I/II molecules allows MSCs to evade the host immune system, reducing the risk of immunogenicity to MSCs regardless of donor source (i.e., from patient or healthy donor)[20].
Figure 1. Mechanisms of action of stem cells for neurological diseases. M1: Proinflammatory M1 microglia subtype; M2: anti-inflammatory M2 microglia subtype; ROS: reactive oxygen species; BBB: blood-brain barrier; BCSB: blood-spinal cord-barrier; BDNF: brain-derived neurotrophic factor; GDNF: glial cell line-derived neurotrophic factor; NGF: nerve growth factor; VEGF: vascular endothelial growth factor; NT-3: neurotrophin-3; IL-10: interleukin 10; TGF-β: transforming growth factor beta; IL-1β: interleukin 1-beta; TNF-α: tumor necrosis factor-alpha; MSC: Mesenchymal stromal/stem cell. Created in BioRender. Windsor R. (2026) https://BioRender.com/h3ywdw8.
Across species, MSCs exhibit similar biological properties, including plastic adherence, multipotent differentiation, and shared immunophenotypes. Although there are some species-specific differences in surface markers and immunomodulatory characteristics depending on tissue source, isolation, and culture procedures, the overall biology and therapeutic mechanisms are conserved across species[21]. These similarities support the translational relevance of MSC research across human and veterinary diseases. This review aims to describe the translational potential of stem cell therapy for rare or refractory neurological disorders in humans that share analogous presentations in companion animals [Table 1].
Stem cell therapy applications for human neurological diseases with companion animal correlates
| Human disease | Companion animal disease correlate |
| Fulminant Multiple Sclerosis | Necrotizing Meningoencephalitis in Dogs[34] |
| Primary and Relapsing remitting Multiple Sclerosis | Granulomatous Meningoencephalitis in Dogs[82] |
| Amyotrophic Lateral Sclerosis | Degenerative Myelopathy in Dogs[46,47] |
| Duchenne Muscular Dystrophy | Golden Retriever muscular Dystrophy[51] |
| Viral Encephalitis | Canine Distemper Virus[59,83,84] Feline Infectious Peritonitis[74,75] |
| Myasthenia Gravis | Myasthenia Gravis in Dogs |
| Parkinson’s Disease | Multiple System Degeneration in Kerry Blue Terriers and Chinese Crested Dogs |
| Lennox-Gastaut Syndrome Dravet Syndrome | Refractory Epilepsy in Dogs |
| Leukodystrophies | Canine Globoid Cell Leukodystrophy (Krabbe Disease) |
| Glioblastoma Multiforme | Glioblastoma Multiforme in Dogs[80,81] |
| Alzheimer’s Disease | Canine Cognitive Dysfunction[85] |
| Cerebrovascular Disease | Cerebrovascular Disease in Dogs and Cats |
| Traumatic Brain Injury | Traumatic Brain Injury in Dogs and Cats |
| Spinal Cord Injury | Spinal Cord Injury in dogs[86-90] and Cats |
| Intervertebral Disc Disease | Intervertebral Disc Disease in Dogs[91-94] |
CORRELATIVE DISEASES
Immune-mediated
Multiple sclerosis (humans) and necrotizing meningoencephalitis (dogs)
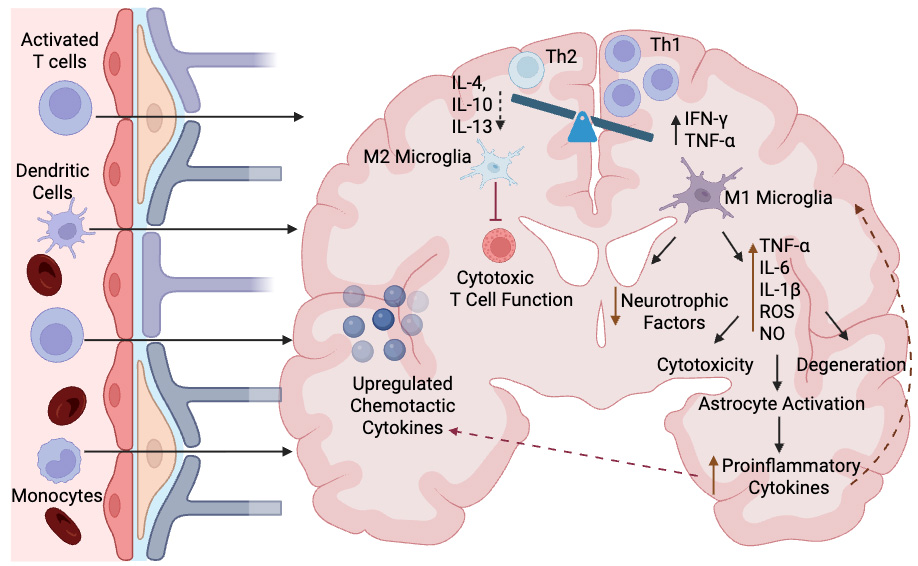
Multiple sclerosis (MS) is a chronic autoimmune disease characterized by demyelination, neuroinflammation, and progressive neurological decline [Figure 2]. MS incidence and phenotype vary across populations, in part due to polymorphism within the MHC, particularly HLA-DRB1 (Human Leukocyte Antigen - DR Beta 1) and HLA-DQB1 (Human Leukocyte Antigen - DQ Beta 1) alleles, which confer susceptibility or protection in an ethnicity-dependent manner[22,23]. Certain variants of MS, such as Marburg type and Baló’s concentric sclerosis, are considered rare, acute forms of the disease affecting only 7%-10% of MS patients[24]. Treatment earlier in the course of disease is generally associated with a slower decline and better overall prognosis[25]. MSC therapy in MS patients has shown promising results, with an estimated 40% of patients improving and 33% remaining stable after therapy[26]. Mechanistic studies have demonstrated increased regulatory T cells, reduced dendritic cell activation, and suppressed lymphocyte proliferation within 24 h of MSC infusion[27]. Functional improvements including enhanced visual evoked potentials and reduced magnetic resonance imaging (MRI) lesion burden have also been documented[28,29].
Figure 2. Pathophysiology of neuroinflammatory disease. Infiltration of peripheral activated T cells, dendritic cells, and monocytes across the blood-brain barrier initiate the transition toward a proinflammatory environment in the brain. This proinflammatory state is dominated by Th1 helper cells, proinflammatory cytokines and M1 proinflammatory microglia. Activation of M1 microglia results in a reduction in neurotrophic factors and increased production of proinflammatory cytokines and other factors which cause cytotoxicity, degeneration and astrocyte activation. Astrocyte activation contributes to further increase in proinflammatory cytokines which stimulate the M1 microglia and the upregulation of chemotactic cytokines to further recruit inflammatory cells into the brain. Reduction in Th2 helper cells, anti-inflammatory cytokines, and M2 microglia reduces the inhibition of cytotoxic T cell function, further perpetuating injury. Created in BioRender. Windsor R (2026) https://BioRender.com/tc9tymv. M1: Proinflammatory M1 microglia subtype; M2: anti-inflammatory M2 microglia subtype; TNF-α: tumor necrosis factor-alpha; IL: interleukin; ROS: reactive oxygen species; IFN-γ: interferon gamma. NO: nitric oxide.
Necrotizing meningoencephalitis (NME) in dogs, seen most commonly in the Pug breed, shares many similarities to the rare fulminant forms of MS including the Marburg variant[30]. Genetic studies have linked NME to dog leukocyte antigen (DLA) class II, paralleling MHC genetic associations in MS[31]. Clinical abnormalities suggestive of an early form of NME mirror the earliest signs of MS in humans[30,32]. Immunological profiling in asymptomatic Pugs genetically at-risk for NME revealed altered T cell subsets and cytokine patterns, suggesting immune imbalance may contribute to the pathogenesis of NME in the Pug[33].
A pilot trial using intravenous (IV) allogeneic MSCs in Pugs demonstrated rapid clinical improvement in all dogs and durable remission with MSC therapy alone for 54% of the dogs (follow-up time 12-43 months)[34]. The shorter lifespan and duration of disease for Pugs with NME compared to the human MS disease course makes the dog a promising animal model to monitor clinical response and relapse rate post MSC therapy in a shorter time window.
While many human MS trials have used intrathecal (IT) and IV autologous MSCs, successful IV allogeneic MSC therapy in Pugs with early NME suggests that IV administration may achieve good central nervous system (CNS) penetration. In addition, allogeneic therapy may offer promising results without evidence of immune reactions. Further research in dogs with NME and potentially granulomatous meningoencephalitis (GME) may help elucidate the short- and long-term effects of MSC therapy in dogs with neuroinflammatory disease. These findings could support the development of MSC therapies for MS in humans.
Myasthenia gravis in humans and dogs
Myasthenia gravis (MG) is a rare autoimmune disorder with a prevalence of 1.5-32.9/100,000 people[35], characterized by antibody-mediated loss of acetylcholine receptors (AChRs) at the neuromuscular junction (NMJ), resulting in skeletal muscle weakness and fatigability. Conventional immunotherapies offer partial symptomatic relief but rarely achieve long-term remission.
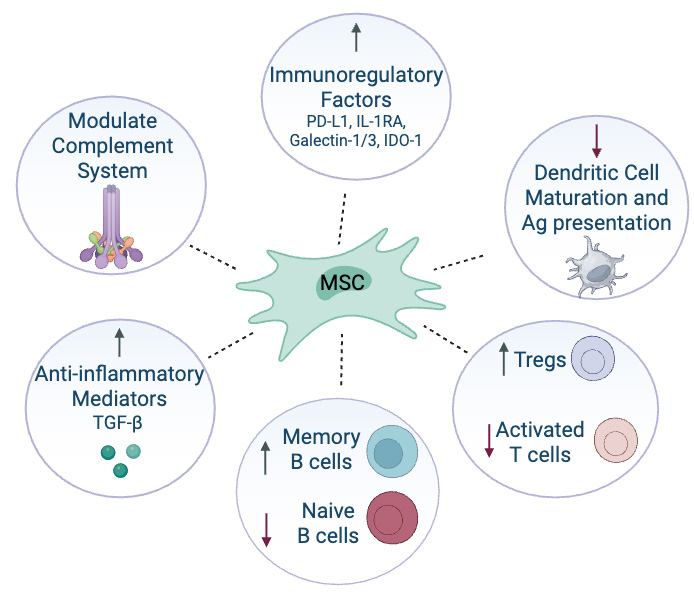
MSCs have emerged as a promising regenerative approach for MG given their immunomodulatory and reparative properties [Figure 3]. Experimental autoimmune MG (EAMG) was first induced in a rabbit in 1973, and since then MG has been induced in several species, including rodents and monkeys[36]. In EAMG rodent models, MSC transplantation restores T helper cell subset 17 (Th17)/regulatory T cell (T reg) balance, reduces anti-AChR antibody titers, and enhances muscle strength through suppression of autoimmune activity and structural restoration of NMJ integrity[37]. These effects are mediated largely through paracrine secretion of growth and trophic factors including vascular endothelial growth factor (VEGF), hepatocyte growth factor (HGF), and insulin-like growth factor 1 (IGF-1) that promote angiogenesis, muscle regeneration, and nerve repair[37]. MSC-derived extracellular vesicles (EVs) have been shown to enhance myoblast fusion and immune clearance of damaged myofibers[37].
Figure 3. Mechanisms of action of stem cell therapy for myasthenia gravis. PD-L1: Programmed death ligand 1; IL-1RA: interleukin-1 receptor antagonist; IDO-1: indoleamine 2,3-dioxygenase 1; T regs: regulatory T cells; TGF-β: transforming growth factor beta; MSC: Mesenchymal stromal/stem cell. Created in BioRender. Windsor R (2026) https://BioRender.com/aqu0egm.
Naturally occurring canine MG is a valuable translational model for testing MSC therapies given the close immunological and clinical resemblance to human MG. Dogs spontaneously develop an autoimmune response against AChRs that mirrors human antibody specificity, particularly targeting the main immunogenic region of the α-subunit and other epitopes on γ subunits[38]. As in humans, canine MG manifests in variable clinical forms (focal, generalized, and fulminant), involves complement-mediated receptor destruction and thymic pathology, and responds to immunosuppression[39]. Unlike induced rodent models, spontaneous canine MG maintains a self-sustaining autoimmune response to better study initiation and remission phases[39]. This makes it uniquely suited for evaluating how MSCs recalibrate immune tolerance and promote long-term NMJ repair under naturally fluctuating conditions. Combining insights from human and canine studies could speed up the development of MSC therapies that offer long-term immune regulation and improved neuromuscular function in MG. A study evaluating the use of MSCs in dogs with MG was completed at the University of California, Davis, with anecdotal reports of success (https://www.vetmed.ucdavis.edu/news/novel-treatment-leads-dogs-recovery) but no published data.
Degenerative disease
Amyotrophic lateral sclerosis (humans) and degenerative myelopathy (dogs)
Amyotrophic lateral sclerosis (ALS) is a fatal neurodegenerative disease marked by motor neuron loss and progressive paralysis. Patients manifest generalized weakness (spinal onset) or difficulty speaking or swallowing (bulbar onset) that typically leads to respiratory failure and death within 2-4 years of diagnosis[40]. ALS is familial in only 10% of patients (fALS), in whom the disease has a strong genetic basis, with 50%-85% of patients exhibiting at least one identified genetic mutation[41]. Superoxide dismutase 1 (SOD1) was the first reported gene mutation in 1993, and since then over 30 additional ALS genes have been identified[41]. MSC therapy has been evaluated in several clinical trials for patients with ALS, showing general safety and modest slowing of disease progression[42]. Proposed mechanisms include neuroprotection, immune modulation, and trophic support.
Advances in the molecular genetics of ALS have made animal disease models particularly promising, with a few targeted molecular therapies currently under development[41]. Degenerative myelopathy (DM) in dogs is genetically and pathologically analogous to familial ALS[43,44]. A missense mutation in the SOD1 gene (E40K) has been identified in DM, with spinal histopathology showing lateral white matter degeneration and SOD1-positive aggregates[43,45].
MSC therapy in DM has demonstrated feasibility and clinical benefit through a variety of administration routes. MRI-guided intra-arterial delivery of radiolabeled MSCs in dogs with DM demonstrated MSC transplantation in the brain and reduction in inflammatory markers[46]. A blinded placebo-controlled trial combining IT allogeneic MSCs with neurorehabilitation extended survival and improved mobility in non-ambulatory dogs with suspected DM (homozygous for SOD1 gene mutation exon 2)[47]. DM may serve as a promising naturally occurring model for ALS, allowing real-world evaluation of MSC delivery methods and functional outcomes.
Inherited
Duchenne muscular dystrophy (humans) and golden retriever muscular dystrophy (dogs)
Duchenne muscular dystrophy (DMD) is a rare X-linked disorder caused by mutations in the dystrophin gene, resulting in loss of dystrophin, sarcolemmal instability, myofiber necrosis, chronic inflammation, and progressive replacement of muscle with fat and fibrotic tissue. The absence of dystrophin disrupts the connection between the cytoskeleton and extracellular matrix within the dystrophin-glycoprotein complex, producing membrane fragility and contraction-induced injury[48]. Over time, repeated cycles of degeneration exhaust regenerative satellite cell pools, resulting in severe muscle weakness, respiratory compromise, and cardiomyopathy. This pathophysiology is consistently observed across species; however, humans and dystrophic dogs exhibit more severe progressive disease, while mdx mice display milder phenotypes, limiting translational accuracy[49]. Golden Retriever muscular dystrophy (GRMD) closely recapitulates human disease, exhibiting early-onset weakness, progressive fibrosis, respiratory impairment, and cardiomyopathy[48].
Stem-cell-based therapies aim to restore muscle function through regeneration, dystrophin replacement, and modulation of the inflammatory environment. Intravenous injection of human adipose-derived MSCs into four 2-month-old dogs with GRMD demonstrated that MSCs were able to reach, engraft, and express human dystrophin in dystrophic muscle up to six months after transplantation[50]. Human immature dental pulp stem cells successfully migrated to and engrafted within GRMD dog muscle, producing modest dystrophin expression and showing greater clinical benefit when delivered systemically and repeatedly rather than by single local injection[51]. These studies have helped set the stage for pilot trials using MSCs in patients with Becker muscular dystrophy (BMD) and DMD that demonstrated increased creatinine kinase, increased dystrophin-positive levels and muscle fibers, and variable improvement in limb strength and gait[52,53].
Altogether, stem cell therapies represent a compelling but still developing avenue, with large-animal models, particularly GRMD dogs, serving as critical translational bridges between laboratory findings and human clinical application. Some dogs with GRMD and other DMD gene mutations live well into adulthood. Owners have typically developed a strong bond with these companion dogs and are open to treatment strategies, including MSCs and forms of gene therapy (Joe Kornegay, personal communication). However, treatment availability and cost limit access to these novel treatments. Clinical trials evaluating stem cell therapy in these valuable translational models could greatly benefit GRMD patients and humans alike.
Lennox-gastaut syndrome and dravet syndrome (humans) and refractory epilepsy (dogs)
Rare refractory childhood epileptic syndromes including Lennox-Gastaut Syndrome (LGS) and Dravet Syndrome (DS) are marked by frequent and severe seizures, developmental delays, and resistance to standard therapies[54]. The incidence and prevalence of these syndromes vary based on study and geographical location; however, DS is rarer, affecting 2.17-6.5 per 100,000 people, compared with 1.9-60.8 per 100,000 people for LGS[54,55]. Canine epilepsy affects 0.6%-0.75% of dogs and presents with similar seizure types, comorbidities, and pharmacoresistance[56]. Attention deficits, mood disturbances, and sleep disorders are common in affected children and dogs, reflecting the broad impact of chronic refractory epilepsy on brain function[56].
Refractory epilepsy is characterized by underlying neuroinflammation, altered neurotransmitter signaling, and disrupted neuronal networks, which contribute to seizure persistence and pharmacoresistance [Figure 4]. MSCs are being explored in human patients with refractory epilepsy and may offer new hope for patients with poor response to conventional drugs. MSCs may reduce seizure frequency by modulating neuroinflammation and restoring neuronal balance. IT and IV infusions appear safe, with ongoing studies exploring optimal dosing and timing[57]. Studies in humans have shown preliminary efficacy in reducing seizure burden, improving electroencephalography (EEG) markers, and reducing comorbid anxiety and depression[57]. Animal models evaluating MSC therapy for epilepsy have used predominantly rodents[58], although MSC therapy has led to durable seizure reduction in canine pilot studies of dogs with seizure disorders secondary to canine distemper virus (CDV) and NME[34,59]. Canine refractory epileptics may prove a valuable model for optimizing MSC therapeutic strategies and evaluating long-term efficacy, offering a natural disease pathology that more closely parallels refractory childhood epilepsy than induced rodent models.
Globoid cell leukodystrophy (Krabbe Disease) (humans and dogs)
Leukodystrophies comprise a heterogeneous group of inherited disorders characterized by progressive dysfunction of CNS myelin. Overall, leukodystrophies affect 1 in 7,663 live births in the United States[60]; however, individual diseases are rare[61], including Krabbe Disease (KD), which affects 0.3-2.6 per 100,000 live births[62]. KD is an autosomal recessive lysosomal disorder caused by variants in the GALC gene that reduce galactosylceramidase (GALC) activity, leading to psychosine accumulation, cerebral white matter degeneration, and peripheral neuropathy[62].
Animal models remain essential for understanding disease mechanisms and developing new therapies. Rodent models and zebrafish have enabled detailed dissection of oligodendrocyte biology, lysosomal dysfunction, and inflammatory responses, yet their small brain size, rapid disease progression, and limited white-matter complexity restrict their translational value for testing regenerative or gene-augmented cell therapies. Large-animal models including canine globoid cell leukodystrophy/KD offer a critical intermediate step between rodents and humans, as affected dogs naturally exhibit clinically and pathologically authentic demyelination, peripheral neuropathy, and long-tract degeneration in a brain more comparable in size, myelination pattern, and immune structure to the human CNS[63].
Recent advances in stem-cell and gene-modified stem-cell therapies have demonstrated therapeutic promise in leukodystrophies such as metachromatic leukodystrophy (MLD), X-linked adrenoleukodystrophy (X-ALD), and KD. These approaches aim to provide cross-correcting enzymes, promote remyelination, modulate neuroinflammation, or replace dysfunctional glia. While most preclinical work has been performed in rodents, large-animal studies including adeno-associated virus (AAV)-based and cell-based interventions in canine KD have shown that dogs can exhibit clinically meaningful improvement, guide vector optimization, and reveal treatment-related toxicities not evident in small animals. Because stem cell therapies depend critically on cell migration, engraftment, and long‐distance myelin repair within a large CNS environment, the canine globoid cell leukodystrophy model may be a more predictive platform for advancing stem-cell therapies toward human clinical trials[63].
Infectious
Viral encephalitis (humans) and distemper virus (dogs) and feline infectious peritonitis (cats)
Infectious encephalitis is generally rare in humans, with incidences ranging between 1.4-7 cases/100,000 people per year[64,65]. Infectious encephalitis is often associated with a guarded prognosis, with fatality rate as high as 18% and severe debilitation in more than half of long-term survivors[66]. Viral infections account for approximately 70% of confirmed cases of encephalitis and can result from direct viral invasion or secondary immune-mediated damage[67].
Coronavirus disease 2019 (COVID-19) is caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus. It has been increasingly recognized for its impact beyond the respiratory system, with a growing body of evidence highlighting its neurological complications. Patients with COVID-related encephalitis may experience seizures, confusion, altered consciousness, and long-term cognitive deficits. The virus can disrupt the blood-brain barrier, activate microglia and astrocytes, and trigger a cascade of pro-inflammatory cytokines, leading to neuroinflammation and neuronal injury[68]. In some cases, neurological symptoms persist long after the acute infection has resolved, contributing to what is now referred to as “long COVID”. These post-viral syndromes often include brain fog, memory impairment, and mood disturbances, suggesting ongoing immune dysregulation within the CNS[68].
The complexity and persistence of these neurological effects underscore the need for therapies that can modulate inflammation and support neural recovery. The immunoregulatory and neuroprotective effects of MSCs and MSC-derived EVs have been described for acute neurological COVID-19; however, clinical efficacy remains under investigation[68]. MSC therapy has also been explored as a treatment for Japanese encephalitis virus (JEV), a leading cause of viral encephalitis in Asia[69]. MSC treatment significantly reduced mortality and improved recovery in JEV-infected mice by attenuating inflammatory responses, microglia activation, neuronal injury, blood-brain barrier disruption, and viral load[69]. In vitro, MSCs promoted microglial polarization from M1 (pro-inflammatory) to M2 (anti-inflammatory) phenotypes, enhanced neuronal survival, and induced interferon-α/β-mediated antiviral effects[69].
CDV causes demyelinating leukoencephalitis with glial activation and cytokine dysregulation, paralleling human viral encephalitides[70,71]. Differing neurological syndromes have been reported with acute and chronic CDV, mirroring acute viral encephalitis including measles and acute COVID and post-viral neuroinflammation associated with long COVID. MSC therapy in CDV infection has shown mixed results. In a small study of CDV dogs, MSCs did not improve survival in dogs with acute disease; however, intra-arterial MSCs reduced myoclonus and restored ambulation in three of four dogs with chronic CDV[72]. A larger retrospective study reported improved ambulation and reduced seizures and myoclonus after IV allogeneic MSC therapy in 10 of 14 dogs, with sustained improvement during the six-month follow-up[59].
Although dogs are the stronger translational model for most neurological conditions, feline infectious peritonitis (FIP) serves as a promising viral translational model in the cat. FIP arises when a typically benign feline enteric coronavirus mutates within white blood cells, triggering a systemic and often fatal immune-mediated inflammatory response in tissues around vessels in organs such as the abdomen, kidneys, and brain.
Recent studies demonstrate that treatment with the antiviral GS-441524 achieves an overall success rate of approximately 84%-86% in affected cats, significantly improving survival and clinical outcomes[73]. The standard GS-441524 treatment protocol for FIP spans 84 days, which can be financially and logistically challenging for some pet owners. This has prompted growing interest in adjunctive immunomodulatory therapies that could potentially reduce the duration of treatment. In one recent study evaluating cats with effusive FIP, MSCs combined with GS-441524 increased regulatory T cells, reduced exhaustion markers, and improved recovery, demonstrating MSC-mediated immune normalization in a coronavirus-associated CNS disease[74]. In another case series, three cats with FIP were treated exclusively with MSC therapy without any antiviral medication; all three achieved full remission within 21 days of treatment[75]. The functional benefits observed in these naturally occurring viral neuroinflammatory diseases in companion animals offer platforms for studying MSC-mediated neuroprotection and immune regulation in viral encephalitis.
Neoplastic
Glioblastoma multiforme (humans and dogs)
Glioblastoma multiforme (GBM) is an aggressive brain tumor affecting 0.59 to 5 per 100,000 people with a median survival time of ten months after diagnosis[76]. MSC‐based therapy has emerged as a promising strategy for glioma treatment due to the innate tumor-tropism, immunomodulatory capacity, and engineering versatility of MSCs. Native MSCs can exert intrinsic anti-glioma effects by limiting angiogenesis, inducing apoptosis, and suppressing tumor cell proliferation. These processes are linked to downregulation of platelet-derived growth factor (PDGF)/platelet-derived growth factor receptor (PDGFR) signaling, inhibition of focal adhesion kinase (FAK) and integrin pathways, and decreased VEGF production within the tumor microenvironment[77]. MSCs also modulate glioma cell cycling by reducing cyclin D1 expression and can sensitize glioblastoma cells to temozolomide (TMZ), enhancing chemotherapeutic efficacy[77].
Beyond these native effects, MSCs serve as potent cellular vehicles for targeted delivery of anti-cancer agents. These include chemotherapeutics (e.g., paclitaxel, doxorubicin), therapeutic genes [e.g., Bone Morphogenetic Protein 4 (BMP4), Recombinant carboxylesterase enzyme (rCE) for Irinotecan (CPT-11) activation], pro-apoptotic ligands [e.g., Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand (TRAIL)], immunomodulatory cytokines [e.g., interleukin (IL)-7, IL-12], and oncolytic viruses, resulting in reduced tumor volume and prolonged survival in preclinical glioma models[77]. Their migratory capacity allows MSCs to deliver cytotoxic or immunostimulatory cargo deep within infiltrative glioma tissue, overcoming limitations of conventional drug penetration.
Spontaneously arising gliomas in companion dogs closely mirror the biological, histopathologic, and clinical features of human gliomas, making dogs a powerful translational model for studying glioma pathogenesis and testing novel therapies. Canine gliomas exhibit comparable tumor morphology, infiltrative behavior, and immunohistochemical expression patterns, with marker intensity correlating to tumor grade in ways that parallel human disease[78]. MRI characteristics in dogs also resemble those of human gliomas, which improves cross-species comparability of diagnostic imaging[78]. Importantly, unlike many rodent models, dogs develop gliomas spontaneously within an immunocompetent and genetically diverse background and share similar genetic drivers and epigenetic features, further strengthening their translational relevance[79].
Clinical trials evaluating canine MSCs in animal models and MSC therapies in canine glioma models show promising results. Ten dogs diagnosed with high-grade rostrotentorial gliomas received eight weekly intravenous doses of dCelyvir, a drug containing MSCs loaded with the oncolytic adenovirus ICOCAV17 (a genetically engineered oncolytic adenovirus)[80]. dCelyvir was well tolerated, resulted in partial response in two dogs and stable disease in three dogs, showed systemic and occasional intratumoral viral detection, and was associated with immune microenvironment shifts linked to response[80]. Canine adipose-derived MSCs engineered to express thymidine kinase were evaluated using in vitro ganciclovir and an in vivo murine U87 glioblastoma model[81]. The modified cells preserved MSC characteristics, altered their secretory and exosomal profiles toward antitumor immune signaling, and demonstrated clear antitumor efficacy in vitro and in vivo, supporting their potential as a translational gene therapy platform for glioma[81]. The canine glioma model may serve as an invaluable platform for bridging the gap between preclinical studies and human clinical trials evaluating stem cell therapy for GBM.
CONCLUSION
Stem cell therapy represents a transformative approach to treating neurological diseases. MSCs have shown potential to modulate inflammation, promote repair, and improve clinical outcomes across a range of conditions. Studies in dogs laid the groundwork for stem cell therapy in human conditions, and spontaneous animal models offer valuable platforms for testing and refining MSC therapies to accelerate translational research. Naturally occurring diseases in companion animals provide real-world insights into disease progression, therapeutic response, and patient variability. These models can inform human trial design, biomarker development, and endpoint selection. By aligning human and veterinary research, we can accelerate the development of effective treatments for rare, complex, and challenging neurological diseases across species.
DECLARATIONS
Acknowledgments
I would like to express my sincere gratitude to Dr. Will Pass for his assistance in reference acquisition and organization.
Authors’ contributions
The author contributed solely to the article.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
Dr. Windsor R is an employee of Gallant and serves as the Director of Veterinary Affairs.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Gunes D, Karaca M, Durmus A, et al. Challenges in the clinical management of rare diseases and center-based multidisciplinary approach to creating solutions. Eur J Pediatr. 2025;184:281.
2. Dani KA, Murray LJ, Razvi S. Rare neurological diseases: a practical approach to management. Pract Neurol. 2013;13:219-27.
3. Shan Z, Ding L, Zhu C, Sun R, Hong W. Medical care of rare and undiagnosed diseases: prospects and challenges. Fundam Res. 2022;2:851-8.
4. Hamasaki MY, Machado MCC, Pinheiro da Silva F. Animal models of neuroinflammation secondary to acute insults originated outside the brain. J Neurosci Res. 2018;96:371-8.
5. Hong H, Kim BS, Im HI. Pathophysiological role of neuroinflammation in neurodegenerative diseases and psychiatric disorders. Int Neurourol J. 2016;20 Suppl 1:S2-7.
6. Gurda BL, Bradbury AM, Vite CH. Canine and feline models of human genetic diseases and their contributions to advancing clinical therapies. Yale J Biol Med. 2017;90:417-31.
7. Zhao H, Liu E, Zhang YQ. Dog models of human atherosclerotic cardiovascular diseases. Mamm Genome. 2023;34:262-9.
8. Karkamo V, Airas N, Lindén J, et al. Severe spontaneous atherosclerosis in two korat breed cats is comparable to human atherosclerosis. J Comp Pathol. 2021;188:52-61.
9. Hytönen MK, Arumilli M, Lappalainen AK, et al. Molecular characterization of three canine models of human rare bone diseases: Caffey, van den Ende-Gupta, and Raine Syndromes. PLoS Genet. 2016;12:e1006037.
10. Kol A, Arzi B, Athanasiou KA, et al. Companion animals: translational scientist’s new best friends. Sci Transl Med. 2015;7:308ps21.
11. Hoang DM, Pham PT, Bach TQ, et al. Stem cell-based therapy for human diseases. Signal Transduct Target Ther. 2022;7:272.
12. Graves SS, Storb R. Evolution of haematopoietic cell transplantation for canine blood disorders and a platform for solid organ transplantation. Vet Med Sci. 2021;7:2156-71.
13. Graves SS, Storb R. Developments and translational relevance for the canine haematopoietic cell transplantation preclinical model. Vet Comp Oncol. 2020;18:471-83.
14. Lupu M, Storb R. Five decades of progress in haematopoietic cell transplantation based on the preclinical canine model. Vet Comp Oncol. 2007;5:14-30.
15. Pittenger MF, Discher DE, Péault BM, Phinney DG, Hare JM, Caplan AI. Mesenchymal stem cell perspective: cell biology to clinical progress. NPJ Regen Med. 2019;4:22.
16. Voga M, Adamic N, Vengust M, Majdic G. Stem cells in veterinary medicine-current state and treatment options. Front Vet Sci. 2020;7:278.
17. Han X, Liao R, Li X, et al. Mesenchymal stem cells in treating human diseases: molecular mechanisms and clinical studies. Signal Transduct Target Ther. 2025;10:262.
18. Acosta SA, Tajiri N, Hoover J, Kaneko Y, Borlongan CV. Intravenous bone marrow stem cell grafts preferentially migrate to spleen and abrogate chronic inflammation in stroke. Stroke. 2015;46:2616-27.
19. Jung HS, Jeong SY, Yang J, et al. Neuroprotective effect of mesenchymal stem cell through complement component 3 downregulation after transient focal cerebral ischemia in mice. Neurosci Lett. 2016;633:227-34.
20. Ichiryu N, Fairchild PJ. Immune privilege of stem cells. In: Zavazava N, Editors. Embryonic Stem Cell Immunobiology. Humana Press, Totowa, NJ; 2013. pp. 1-16.
21. Uder C, Brückner S, Winkler S, Tautenhahn HM, Christ B. Mammalian MSC from selected species: features and applications. Cytometry A. 2018;93:32-49.
22. Fguirouche A, Naji Y, Guennouni M, et al. HLA-DRB1 and DQB1 allelic polymorphism and multiple sclerosis in a moroccan population. Curr Issues Mol Biol. 2025;47.
23. Kim W, Patsopoulos NA. Genetics and functional genomics of multiple sclerosis. Semin Immunopathol. 2022;44:63-79.
24. Capello E, Mancardi GL. Marburg type and Balò’s concentric sclerosis: rare and acute variants of multiple sclerosis. Neurol Sci. 2004;25 Suppl 4:S361-3.
25. Singer BA, Feng J, Chiong-Rivero H. Early use of high-efficacy therapies in multiple sclerosis in the United States: benefits, barriers, and strategies for encouraging adoption. J Neurol. 2024;271:3116-30.
26. Islam MA, Alam SS, Kundu S, et al. Mesenchymal stem cell therapy in multiple sclerosis: a systematic review and meta-analysis. J Clin Med. 2023;12:6311.
27. Karussis D, Karageorgiou C, Vaknin-Dembinsky A, et al. Safety and immunological effects of mesenchymal stem cell transplantation in patients with multiple sclerosis and amyotrophic lateral sclerosis. Arch Neurol. 2010;67:1187-94.
28. Connick P, Kolappan M, Crawley C, et al. Autologous mesenchymal stem cells for the treatment of secondary progressive multiple sclerosis: an open-label phase 2a proof-of-concept study. Lancet Neurol. 2012;11:150-6.
29. Petrou P, Kassis I, Levin N, et al. Beneficial effects of autologous mesenchymal stem cell transplantation in active progressive multiple sclerosis. Brain. 2020;143:3574-88.
30. Windsor R, Stewart S, Huentelman M, Keller S, Khanna C. An early clinical phenotype of necrotizing meningoencephalitis in the Pug reveals similarities to multiple sclerosis in humans. Am J Vet Res. 2024;85.
31. Greer KA, Wong AK, Liu H, et al. Necrotizing meningoencephalitis of Pug dogs associates with dog leukocyte antigen class II and resembles acute variant forms of multiple sclerosis. Tissue Antigens. 2010;76:110-8.
32. Windsor R, Stewart S, Schmidt J, et al. A potential early clinical phenotype of necrotizing meningoencephalitis in genetically at-risk pug dogs. J Vet Intern Med. 2022;36:1382-9.
33. Windsor R, Stewart SD, Talboom J, et al. Leukocyte and cytokine variables in asymptomatic Pugs at genetic risk of necrotizing meningoencephalitis. J Vet Intern Med. 2021;35:2846-52.
34. Windsor RC, Stewart S, Cassano JM, et al. Intravenous allogeneic mesenchymal stromal cell therapy in 13 Pugs with presumptive early necrotizing meningoencephalitis. J Am Vet Med Assoc. 2025;263:1-10.
35. Ye Y, Murdock DJ, Chen C, Liedtke W, Knox CA. Epidemiology of myasthenia gravis in the United States. Front Neurol. 2024;15:1339167.
36. Mantegazza R, Cordiglieri C, Consonni A, Baggi F. Animal models of myasthenia gravis: utility and limitations. Int J Gen Med. 2016;9:53-64.
37. Zhang X, Zhang D, Zhang Y, Wang J, Lu J. Restoration of skeletal muscle function via mesenchymal stem cells: mechanistic insights and therapeutic advances in myasthenia gravis. Front Cell Dev Biol. 2025;13:1658062.
38. Shelton GD, Cardinet GH, Lindstrom JM. Canine and human myasthenia gravis autoantibodies recognize similar regions on the acetylcholine receptor. Neurology. 1988;38:1417-23.
39. Shelton GD. Acquired myasthenia gravis: what we have learned from experimental and spontaneous animal models. Vet Immunol Immunopathol. 1999;69:239-49.
40. Longinetti E, Fang F. Epidemiology of amyotrophic lateral sclerosis: an update of recent literature. Curr Opin Neurol. 2019;32:771-6.
41. Nijs M, Van Damme P. The genetics of amyotrophic lateral sclerosis. Curr Opin Neurol. 2024;37:560-9.
42. Morata-Tarifa C, Azkona G, Glass J, Mazzini L, Sanchez-Pernaute R. Looking backward to move forward: a meta-analysis of stem cell therapy in amyotrophic lateral sclerosis. NPJ Regen Med. 2021;6:20.
43. Awano T, Johnson GS, Wade CM, et al. Genome-wide association analysis reveals a SOD1 mutation in canine degenerative myelopathy that resembles amyotrophic lateral sclerosis. Proc Natl Acad Sci U S A. 2009;106:2794-9.
44. Zeng R, Coates JR, Johnson GC, et al. Breed distribution of SOD1 alleles previously associated with canine degenerative myelopathy. J Vet Intern Med. 2014;28:515-21.
45. Crisp MJ, Beckett J, Coates JR, Miller TM. Canine degenerative myelopathy: biochemical characterization of superoxide dismutase 1 in the first naturally occurring non-human amyotrophic lateral sclerosis model. Exp Neurol. 2013;248:1-9.
46. Malysz-Cymborska I, Golubczyk D, Kalkowski L, et al. Intra-arterial transplantation of stem cells in large animals as a minimally-invasive strategy for the treatment of disseminated neurodegeneration. Sci Rep. 2021;11:6581.
47. Gouveia D, Correia J, Cardoso A, et al. Intensive neurorehabilitation and allogeneic stem cells transplantation in canine degenerative myelopathy. Front Vet Sci. 2023;10:1192744.
48. Kornegay JN. The golden retriever model of Duchenne muscular dystrophy. Skelet Muscle. 2017;7:9.
49. Gaina G, Popa Gruianu A. Muscular dystrophy: experimental animal models and therapeutic approaches (review). Exp Ther Med. 2021;21:610.
50. Vieira NM, Valadares M, Zucconi E, et al. Human adipose-derived mesenchymal stromal cells injected systemically into GRMD dogs without immunosuppression are able to reach the host muscle and express human dystrophin. Cell Transplant. 2012;21:1407-17.
51. Kerkis I, Ambrosio CE, Kerkis A, et al. Early transplantation of human immature dental pulp stem cells from baby teeth to golden retriever muscular dystrophy (GRMD) dogs: Local or systemic? J Transl Med. 2008;6:35.
52. Li P, Cui K, Zhang B, et al. Transplantation of human umbilical cord-derived mesenchymal stems cells for the treatment of Becker muscular dystrophy in affected pedigree members. Int J Mol Med. 2015;35:1051-7.
53. Dai A, Baspinar O, Yeşilyurt A, et al. Efficacy of stem cell therapy in ambulatory and nonambulatory children with Duchenne muscular dystrophy - Phase I-II. Degener Neurol Neuromuscul Dis. 2018;8:63-77.
54. Sullivan J, Benítez A, Roth J, et al. A systematic literature review on the global epidemiology of Dravet syndrome and Lennox-Gastaut syndrome: prevalence, incidence, diagnosis, and mortality. Epilepsia. 2024;65:1240-63.
55. Strzelczyk A, Zuberi SM, Striano P, Rosenow F, Schubert-Bast S. The burden of illness in Lennox-Gastaut syndrome: a systematic literature review. Orphanet J Rare Dis. 2023;18:42.
57. Ramos-Fresnedo A, Perez-Vega C, Domingo RA, et al. Mesenchymal stem cell therapy for focal epilepsy: a systematic review of preclinical models and clinical studies. Epilepsia. 2022;63:1607-18.
58. Altalhi AS, Javaid MS, Jones NC, et al. Effects of cell therapy on seizures in animal models of epilepsy: systematic review and meta-analysis. Epilepsia. 2026;67:13-26.
59. Brunel HDSS, Villaroel CL, Dallago BSL, Pogue R, Malard PF. Retrospective study of mesenchymal stem cell therapy in dogs with neurological complications resulting from infection by canine distemper virus. Braz. J. Sci. 2022;1:73-81.
60. Bonkowsky JL, Nelson C, Kingston JL, Filloux FM, Mundorff MB, Srivastava R. The burden of inherited leukodystrophies in children. Neurology. 2010;75:718-25.
61. Mahdieh N, Soveizi M, Tavasoli AR, et al. Genetic testing of leukodystrophies unraveling extensive heterogeneity in a large cohort and report of five common diseases and 38 novel variants. Sci Rep. 2021;11:3231.
62. Ream MA, Lam WKK, Grosse SD, et al. Evidence and recommendation for infantile Krabbe Disease newborn screening. Pediatrics. 2025;155:e2024069152.
63. Rutherford HA, Hamilton N. Animal models of leukodystrophy: a new perspective for the development of therapies. FEBS J. 2019;286:4176-91.
64. Boucher A, Herrmann JL, Morand P, et al. Epidemiology of infectious encephalitis causes in 2016. Med Mal Infect. 2017;47:221-35.
65. Duerlund LS, Nielsen H, Bodilsen J. Current epidemiology of infectious encephalitis: a narrative review. Clin Microbiol Infect. 2025;31:515-21.
66. Zhao W, Zhou Y, Hu Y, et al. Predictors of mortality and poor outcome for patients with severe infectious encephalitis in the intensive care unit: a cross-sectional study. BMC Infect Dis. 2024;24:421.
67. Said S, Kang M. Viral encephalitis. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025. Available from: https://www.ncbi.nlm.nih.gov/books/NBK470162/. [Last accessed on 20 Apr 2026].
68. León-Moreno LC, Reza-Zaldívar EE, Hernández-Sapiéns MA, et al. Mesenchymal stem cell-based therapies in the post-acute neurological COVID syndrome: current landscape and opportunities. Biomolecules. 2023;14:8.
69. Bian P, Ye C, Zheng X, et al. Mesenchymal stem cells alleviate Japanese encephalitis virus-induced neuroinflammation and mortality. Stem Cell Res Ther. 2017;8:38.
70. Lempp C, Spitzbarth I, Puff C, et al. New aspects of the pathogenesis of canine distemper leukoencephalitis. Viruses. 2014;6:2571-601.
71. Rendon-Marin S, da Fontoura Budaszewski R, Canal CW, Ruiz-Saenz J. Tropism and molecular pathogenesis of canine distemper virus. Virol J. 2019;16:30.
72. Pinheiro LL, de Lima AR, Martins DM, et al. Mesenchymal stem cells in dogs with demyelinating leukoencephalitis as an experimental model of multiple sclerosis. Heliyon. 2019;5:e01857.
73. Gokalsing E, Ferrolho J, Gibson MS, Vilhena H, Anastácio S. Efficacy of GS-441524 for feline infectious peritonitis: a systematic review (2018-2024). Pathogens. 2025;14:717.
74. Wanakumjorn P, Kimura K, Castillo D, et al. Mesenchymal stem/stromal cell therapy improves immune recovery in a feline model of severe coronavirus infection. Stem Cells Transl Med. 2025;14:szaf025.
75. Mohamadian S, Kazerooni P, Taheri Mirghaed A, et al. ; Department of Clinical Science, Faculty of Veterinary Medicine, Urmia Branch, Islamic Azad University, Urmia, Iran. Successful treatment of feline infectious peritonitis disease (FIP) with mesenchymal stem cells. Iran J Vet Med. 2025;19:397-404.
76. Grech N, Dalli T, Mizzi S, Meilak L, Calleja N, Zrinzo A. Rising incidence of glioblastoma multiforme in a well-defined population. Cureus. 2020;12:e8195.
77. Nowak B, Rogujski P, Janowski M, Lukomska B, Andrzejewska A. Mesenchymal stem cells in glioblastoma therapy and progression: how one cell does it all. Biochim Biophys Acta Rev Cancer. 2021;1876:188582.
78. Herranz C, Fernández F, Martín-Ibáñez R, et al. Spontaneously arising canine glioma as a potential model for human glioma. J Comp Pathol. 2016;154:169-79.
79. Yost NM, Angelastro JM. Canine glioma as a model for human glioblastoma. In: Agrawal A, Singh Kunwar D, Editors. Glioblastoma-current evidence. IntechOpen, 2022.[DOI:10.5772/intechopen.106464].
80. Cloquell A, Mateo I, Gambera S, et al. Systemic cellular viroimmunotherapy for canine high-grade gliomas. J Immunother Cancer. 2022;10:gmr.15028310.
81. Villatoro AJ, Alcoholado C, Martín-Astorga MDC, et al. Suicide gene therapy by canine mesenchymal stem cell transduced with thymidine kinase in a u-87 glioblastoma murine model: secretory profile and antitumor activity. PLoS ONE. 2022;17:e0264001.
82. Zeira O, Asiag N, Aralla M, et al. Adult autologous mesenchymal stem cells for the treatment of suspected non-infectious inflammatory diseases of the canine central nervous system: safety, feasibility and preliminary clinical findings. J Neuroinflammation. 2015;12:181.
83. Gonçalves D, Gomes M, Guterra V, et al. Research article mesenchymal stem cell infusion for the treatment of neurological sequelae of canine distemper virus: a clinical study. Genet Mol Res. 2018;17:gmr18088.
84. Pinheiro AO, Cardoso MT, Vidane AS, et al. Controversial results of therapy with mesenchymal stem cells in the acute phase of canine distemper disease. Genet Mol Res. 2016;15:gmr.15028310.
85. Kim TY, Kim NH, Chae JA, et al. Evaluation of cognitive and mobility function in geriatric dogs following treatment with stem cell and stem cell extracellular vesicles derived from embryonic stem cells: a pilot study. Front Vet Sci. 2025;12:1549870.
86. Kim Y, Jo SH, Kim WH, Kweon OK. Antioxidant and anti-inflammatory effects of intravenously injected adipose derived mesenchymal stem cells in dogs with acute spinal cord injury. Stem Cell Res Ther. 2015;6:229.
87. Vikartovska Z, Kuricova M, Farbakova J, et al. Stem cell conditioned medium treatment for canine spinal cord injury: pilot feasibility study. Int J Mol Sci. 2020;21:5129.
88. Sharun K, Kumar R, Chandra V, et al. Percutaneous transplantation of allogenic bone marrow-derived mesenchymal stem cells for the management of paraplegia secondary to Hansen type I intervertebral disc herniation in a Beagle dog. Iran J Vet Res. 2021;22:161-6.
89. Orlandin JR, Gomes IDS, Sallum Leandro SF, et al. Treatment of chronic spinal cord injury in dogs using amniotic membrane-derived stem cells: preliminary results. Stem Cells Cloning. 2021;14:39-49.
90. Deng WS, Yang K, Liang B, Liu YF, Chen XY, Zhang S. Collagen/heparin sulfate scaffold combined with mesenchymal stem cells treatment for canines with spinal cord injury: a pilot feasibility study. J Orthop Surg. 2021;29:23094990211012293.
91. Steffen F, Smolders LA, Roentgen AM, Bertolo A, Stoyanov J. Bone marrow-derived mesenchymal stem cells as autologous therapy in dogs with naturally occurring intervertebral disc disease: feasibility, safety, and preliminary results. Tissue Eng Part C Methods. 2017;23:643-51.
92. Steffen F, Bertolo A, Affentranger R, Ferguson SJ, Stoyanov J. Treatment of naturally degenerated canine lumbosacral intervertebral discs with autologous mesenchymal stromal cells and collagen microcarriers: a prospective clinical study. Cell Transplant. 2019;28:201-11.
93. Bach FS, Rebelatto CLK, Fracaro L, et al. Comparison of the efficacy of surgical decompression alone and combined with canine adipose tissue-derived stem cell transplantation in dogs with acute thoracolumbar disk disease and spinal cord injury. Front Vet Sci. 2019;6:383.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].