Wearable monitoring of muscle health across fatigue, injury and recovery in sports
Abstract
Muscle fatigue and injury are the core issues that restrict the improvement of athletes’ competitive performance and the maintenance of sports health for the general population. The traditional muscle health management paradigm is limited by lagging assessment and single indicators, failing to meet the demands of precise training and individualized rehabilitation. With the advances in wearable sensing technology, muscle health management has the potential to transform from the empirical modality to a new data-driven paradigm. However, existing publications either focus on materials innovation and structure design in sensor development, or solely highlight the overlapping physiological mechanisms inducing muscle fatigue and injury. Thus, a comprehensive review presenting insights on the physiological relevance between biological signals fluctuations and muscle health status, the detection mechanisms and functional layouts of wearable sensors to capture these signals, as well as their real-world applications in competitive sports and public fitness is timely needed. Herein, this article systematically reviews the physiological mechanisms of muscle fatigue, injury and repair, with a focus on elaborating the characteristic change patterns of related bioelectrical, biochemical and biomechanical markers in the process. Sensing mechanisms and working layouts of wearable technology are comprehensively summarized. Importantly, corresponding applications in real-world settings associated with improving professional athletic performance and public fitness are proposed, including load monitoring, fatigue evaluation, personalized nutrition management, as well as artificial intelligence (AI)-enabled multimodal fusion. Based on this, future perspectives are envisioned to better aid sports activities and engineer the development of sports science and sports medicine.
Keywords
INTRODUCTION
Skeletal muscle is the core executive tissue of human motor function, and its structural integrity and functional stability directly determine athletic performance, training adaptability and injury susceptibility[1-3]. Exercise-induced muscle fatigue and injury is a common problem in the fields of sports science and sports medicine[4]. It not only leads to a decline in short-term motor function, but may cause long-term sequelae such as chronic strain and fibrosis through a vicious cycle of “fatigue accumulation-repair delay-injury recurrence”, undermining athletes’ competitive level and the health maintenance of the general population during sports[5]. The traditional muscle health management model relies on macroscopic training volume statistics, subjective fatigue assessment and static imaging detection[6,7]. It has limitations in lagging assessment and single indicators, and hence can hardly reflect individual physiological response differences and meet the modern sports science demands in “precise training, early warning and personalized rehabilitation”[3]. In recent years, the rapid development of wearable sensing technology, along with accurate biomechanical analysis, artificial intelligence algorithms and microfluidic technology, have brought exciting opportunities to revolutionize muscle health management[8,9].
Wearable devices based on surface electromyography (sEMG) and electrocardiography (ECG) have achieved real-time capture of neuromuscular activities and central regulatory states[10-12]. Body-worn biomechanical monitoring (e.g., inertial measurement unit, pressure sensing insoles, wearable elastography) can quantify the mechanical load, vertical stiffness during running and jumping, and soft tissue elastic changes in exercise[13-16]. This helps to provide an objective basis for injury risk forecasting and healing process tracking. The sweat-based, multi-index biochemical sensing patch has broken the barriers of traditional invasive, intermittent blood sampling and detection, achieving non-invasive, continuous monitoring of physiological metabolite (e.g., lactic acid, electrolytes, and urea), acute and chronic inflammation (e.g., interleukins), and hydration status[17-19]. Innovative fusions of these technologies have stimulated the transformation of muscle health management from “experience-based” to “data-driven”. It benefits forming a multi-dimensional, closed-loop technical system for competitive sports and public fitness, covering exercise intensity monitoring, body fatigue assessment, injury risk prediction, and training recovery intervention. In spite of the promising opportunity, the majority of existing publications focus on listing popular biomarkers and exemplifying corresponding sensor designs and general applications from the perspective of engineering, or solely highlight the overlapping physiological mechanisms inducing muscle fatigue and injury in a biomedical view[20-24]. Insights from the intersection of biomedicine, engineering and sports science that elaborate the physiological relevance between biological signals fluctuations and muscle health status, the detection mechanisms and functional layouts of wearable sensors to capture these signals, as well as their real-world applications in competitive sports and public fitness are yet to be posed.
For this sake, the current review systematically integrates the physiological mechanisms underlying muscle fatigue, injury and healing, with biomarkers and their changes corresponding to the development of the process [Figure 1]. Next, wearable sensing principles and sensor layouts targeting at these bioelectrical, biochemical and biomechanical indicators are comprehensively summarized. The following section highlights the specific applications of wearable muscle health monitoring in real-world sports settings, ranging from load management, fatigue evaluation and exercise and nutrition prescription. Based on the aforementioned advances, insightful outlooks are proposed regarding technological optimization, data integration, application expansion, and cross-disciplinary collaboration, to precisely engineer the development of sports science and sports medicine.
Figure 1. Representative biomarkers and their physiologically changing patterns in different muscle health conditions, as well as corresponding wearable sensor configurations. RMS: Root mean square; iEMG: integrated electromyography; sEMG: surface electromyography; CK: creatine kinase; Mb: myoglobin; IL-6: Interleukin-6; ISF: interstitial fluid; IMU: inertial measurement unit.
PHYSIOLOGICAL BASIS AND INDICATOR DYNAMICS ACROSS MUSCLE FATIGUE, INJURY AND RECOVERY
Physiological principles across muscle fatigue, injury and recovery
Skeletal muscle is the core tissue that maintains life activities and movement functions[25]. Its core roles include driving body movements, maintaining posture stability, assisting respiratory activities, and dissipating heat via energy metabolism[6]. It is a key support for the normal operation of human physiological functions. Muscle fatigue is broadly defined as the functional failure to maintain the required or expected force (or power output) that hinders the muscle performance overtime at a specific load and is accompanied by the feeling of tiredness[26]. While muscle injury refers to the structural destruction of muscle fibers, tendons or surrounding tissues[5]. Sports muscle injuries are also classified into direct injury and indirect injury, where direct injury is caused by the direct action of external impact and is often manifested as muscle contusion or tear, whilst indirect traumatic muscle injuries are mostly induced by excessive stress or over-stretching of skeletal muscle tissue[6]. It is worth noting that there is a significant overlapping association between muscle injury and fatigue[24]. Although pronounced or prolonged fatigue may increase vulnerability to mechanical injury, fatigue and injury remain biologically distinct states within a continuous spectrum of muscle health: one predominantly functional and reversible, the other structural and often persistent[24].
The healing process of muscle fatigue and injury follows a clear physiological mechanism: the body first eliminates damaged tissue fragments through inflammatory responses[27], then activates muscle satellite cells to initiate muscle fiber regeneration, and precisely regulates cell proliferation and differentiation via cytokines[28]. During this process, energy reserves must be restored and the extracellular matrix reshaped, ultimately achieving comprehensive repair of muscle structural integrity and contraction function[29]. Although within a certain range, muscles have the ability to self-repair after injury, this repair process is regulated by multi-dimensional factors, and the precise regulation of molecular pathways is the core link that supports these theories and affects the repair efficiency[4].
Related physiological indicators
Bioelectrical indicators
During the process of muscle fatigue and injury, sEMG is one of the most relevant and sensitive bioelectric indicators reflecting the functional state of muscles. Its time-domain and frequency-domain characteristics showcase regular changes as the injury progresses[30]. In the early stage, time-domain characteristics are mainly reflected as a significant increase in signal amplitude. As fatigue or injury intensifies, an increased number of motor units are recruited to maintain motor function, leading to more muscle fibers contributing to contraction and thus an overall increase in sEMG amplitude[31]. During the process, changes in core time-domain parameters such as integrated EMG (iEMG) and root mean square (RMS) are particularly significant. Both of them show a continuous increasing trend with the aggravation of fatigue or injury and can be used to quantify the degree of muscle fatigue or injury[32].
As for frequency-domain variations, under the state of muscle fatigue, the conduction of muscle fibers slows down, resulting in a reduced propagation rate of action potentials among muscle fibers, which can be witnessed as the frequency components of sEMG signals shift towards the low-frequency range[33]. This change can be intuitively reflected by frequency domain parameters such as median frequency (MF) and mean power frequency (MPF). During the progression of fatigue or injury, both MF and MPF gradually decrease[33]. While once entering the muscle repair and remodeling period, the characteristic parameters of the sEMG signal gradually return to the normal physiological state. Specifically, the total discharge and discharge intensity of the motor unit tend to stabilize, and the time domain (iEMG and RMS) and frequency domain (MF and MPF) features gradually recover[34].
To further enhance the accuracy of muscle state detection, recent studies have focused on fusion detection of sEMG and ECG. It has been confirmed that the fatigue recognition model constructed based on the fusion features of sEMG-ECG can achieve an average recognition rate of 87.83%[35]. Compared with sEMG-only detection, this fusion strategy not only significantly improves the accuracy of fatigue state recognition, but also optimizes the stability and generalization ability of the model, providing more reliable technical support for the early warning and precise assessment of muscle fatigue or injury[35-38].
Biochemical indicators
During the process of muscle fatigue, injury and subsequent healing, biochemical indicators related to energy metabolism, injury and inflammatory response exhibit regular dynamic changes, which can be used as core biological markers for evaluating injury degree and healing status. The following section systematically classifies related biochemical markers into three major categories, namely energy metabolism indicators, muscle injury markers and inflammatory regulation factors. And the changing pattern and physiological relevance of each indicator are clarified in detail.
(1) Energy metabolism indicators
The balance between energy consumption and synthesis is a core parameter to reflect the degree of muscle fatigue, injury and healing. The energy metabolism indicators mainly include reserved glycogen, lactic acid and lactate dehydrogenase, as well as urea nitrogen.
Lactic acid is a characteristic product of anaerobic glycolysis, and the lactate dehydrogenase (LDH) family is a key enzyme system regulating the conversion of lactic acid to pyruvate. In the early stage of fatigue or injury during high-intensity exercise, the muscle oxygen supply is relatively insufficient, activating the anaerobic glycolytic pathway to induce excessive lactic acid production and accumulation in the blood[39]. Such an elevating level of lactic acid within a short period decreases the pH of body fluids, thereby inhibiting muscle fiber contraction function and further impairing athletic performance[40]. It deserves to note that the proportion of LDH isoenzymes undergoes various adjustments facing enhanced anaerobic metabolism, presenting suppressed activities of LDH1 and LDH2 (biased towards aerobic metabolism), while stimulated activities of LDH3, LDH4 and LDH5 (biased towards anaerobic metabolism)[41]. As exercise ceases and oxygen supply resumes in the repair and remodeling period, blood lactic acid is gradually metabolized and cleared through pathways such as oxidative decomposition and gluconeogenesis. The proportion of LDH isoenzymes simultaneously reverses towards aerobic metabolism, with the activities of LDH1 and LDH2 rebounding and the activities of LDH3, LDH4 and LDH5 decreasing[42]. In this way, through monitoring the lactic acid and LDH isoenzymes dynamics, the transformation of muscle metabolic patterns (anaerobic compensation and aerobic homeostasis) can be accurately determined, and the progress of injury repair can be indirectly evaluated. When the proportion of isoenzymes returns to normal, it indicates that the muscle metabolic function has basically recovered[43].
Blood urea nitrogen (BUN) is the final product of protein and amino acid metabolism. Its level changes reflect the energy metabolism pattern and nitrogen balance status insider the body. The normal reference range for BUN is 2.86-7.14 mmol/L[44]. In prolonged and high-intensity exercises, the body will initiate protein (amino acid) catabolism for energy supply, while nucleotide metabolism intensifies, once the breakdown of sugar and fat fails to meet the energy demands. The bi-phasic effect induces increased BUN level with the extended exercise time and the higher intensity[45,46], making it an important signal to indicate insufficient body energy reserve or fatigue. In the repair stage, protein catabolism and BUN production weaken, as energy supply recovers (glycogen reserves rise). At the same time, the kidney’s excretory function is enhanced, accelerating the clearance of BUN in the blood and gradually bringing its level back to the normal range. Therefore, the downward trend of BUN can serve as an important indicator of the restoration of metabolic balance in the body and the relief of fatigue.
(2) Muscle injury markers
Muscle injury can disrupt the integrity of muscle cell membranes, leading to the release of specific substances from cells into the bloodstream. At the same time, imbalance in protein metabolism can also cause changes in characteristic indicators, mainly including creatine kinase (CK) and myoglobin (Mb)[47].
CK is a key enzyme regulating energy conversion within muscle cells, and Mb is an oxygen-carrying protein within muscle cells. Both exist in the cytoplasm of muscle cells, and their concentration changes in blood directly reflect the integrity of the muscle cell membrane and the degree of damage[48]. CK concentration can quantitatively indicate the severity of injury, where > 5,000 U/L is regarded as severe muscle injury[49]. A study analyzing serum samples from 483 male and 245 female athletes established reference intervals for CK activity of 82-1,083 U/L for males and 47-513 U/L for females[50]. Levels exceeding these thresholds may indicate an increased risk of overexertion or injury. Further, a CK level greater than 2,000 U/L two hours post-exercise is widely used to diagnose pathological conditions such as myopathy (e.g., statin-induced myositis) or rhabdomyolysis[51]. As muscle fibers regenerate and the integrity of muscle cell membrane structure recovers, the release of CK and Mb significantly decreases[26]. Meanwhile, the body metabolizes and clears excessive CK and Mb in the blood through liver and kidney[52]. Therefore, the dynamic change of CK and Mb levels can be effective measures to assess muscle injury severity and the repair process, offering potential guides for modifying the training recovery plan[53].
(3) Inflammatory regulation factors
Inflammatory response is the core physiological process during muscle injury and repair, realized by inflammatory regulation factors mainly represented by the interleukin family (IL-6, IL-1, etc.). Inflammatory factors are signal molecules secreted by immune cells (macrophages, lymphocytes) and parenchymal cells (muscle cells, liver cells), widely involved in tissue damage recognition, repair initiation and immune regulation[54], and closely related to the occurrence and development of muscle fatigue or injury[55]. Muscle fatigue or minor injuries caused by high-intensity exercise can trigger the release of inflammatory factors (such as IL-6 and IL-1), which recruit repair cells (such as satellite cells and macrophages)[6,56] at the injury site, clear necrotic tissue fragments, and create a microenvironment favoring muscle fiber regeneration. At this point, a moderate increase in inflammatory factors is recognized as a positive signal for initiating the repair process[55]. However, if the inflammatory response is overly activated or lasts for too long, excessive inflammatory factors will inhibit the proliferation and differentiation of muscle satellite cells, hinder the muscle fiber regeneration and thus delay muscle function restoration[57]. Therefore, inflammatory factor monitoring (e.g., from increase to a stable state and a subsequent decrease) can provide insights into the inflammation regulation state.
Biomechanical indicators
Muscle strength and stiffness are key biomechanical indicators characterizing the functional state of muscles[40]. The former directly reflects the output capacity of the neuromuscular system, while the latter is related to the structural integrity of the muscle-tendon units. These biomechanical cues jointly provide a core quantitative basis for evaluating the functional status and structural attributes of the muscular system[47].
When muscles are fatigued or injured, the maximum force provided by muscle contraction will decrease[56,58]. Studies have shown that after muscle injury, the decline in muscle strength is significantly positively correlated with the severity of muscle fiber damage[59], enabling muscle strength to quantitatively measure the degree of injury[60]. Further from a microscopic view, fatigue or injury disrupts the mechanical interaction between actin and myosin, resulting in muscle microstructure damage, such as muscle fiber rupture and Z-disc distortion[40]. Consequently, the force transmission during muscle contraction is impeded, and the maximum contractile force is thus decreased[61]. As the activation of muscle satellite cells in healing, muscle fiber regeneration and extracellular matrix remodeling gradually advance by restoring the interaction mechanism between actin and myosin and refining the muscle fiber microstructure. This induces the rising of force generation and transmission efficiency in muscle contraction towards the pre-injury level[62]. Based on this, the rate and extent of muscle strength recovery can serve as the core endpoint indexes for scaling muscle function recovery, thus guiding the evaluation and optimization of the efficacy of the rehabilitation plan, especially for professional athletes.
As for the dynamic variations of muscle stiffness, it drastically soars to a peak value when fatigue or injury takes place[63]. This can be explained by the abnormal internal structures, where either muscle fiber damage or inflammation-induced tissue swelling (accumulated with interstitial fluids and inflammatory mediators) stiffens the muscle with reduced elasticity[58]. After the inflammation subsides, the swelling of muscle tissue gradually relieves by the synergy of transporting, processed and neutralizing the excessive interstitial fluid and inflammatory mediators with body’s drainage and immune systems. In this way, the mechanical property of muscle restores, presenting as a soft and elastic tissue[64,65].
Multimodal interpretation of transitions between muscle states across temporal scales
Muscle fatigue, injury and recovery represent a continuous physiological spectrum rather than strictly separated phases[24]. Currently, there exist no universally accepted quantitative thresholds that allow any indicators alone to definitively classify these processes. Instead, differentiation relies on the interpretation of multimodal physiological patterns, temporal evolution, and the reversibility of functional changes.
Muscle fatigue is primarily characterized as a functional and reversible decline in performance capacity without substantial structural disruption[66]. Reported indicators typically demonstrate transient alterations, such as decreased MF and increased RMS in sEMG, elevated metabolic load reflected by increased lactate levels, and minor short-term changes in muscle stiffness. These signals generally return toward baseline following adequate rest[67]. By integrating neuromuscular, metabolic, and mechanical indicators over short time scales and assessing their rapid reversibility relative to individualized baselines, wearable systems can infer fatigue as a transient functional deviation rather than structural impairment.
Muscle injury involves structural disruption at the myofiber or extracellular matrix level[59]. In contrast to fatigue, injury is often associated with persistent strength loss, increased muscle stiffness, prolonged inflammation, and elevations in biochemical markers (such as CK) that indicate muscle membrane damage[68]. Existing biomarkers may reveal indirect manifestations of injury through abnormal motor unit recruitment patterns, sustained asymmetry in movement biomechanics, reduced contractile efficiency, and prolonged deviations from baseline functional metrics[69]. When multimodal signals demonstrate cross-domain abnormalities that persist across extended time scales and fail to recover following rest, it may suggest transition from functional fatigue to structural injury.
Muscle recovery or healing reflects a dynamic remodeling process characterized by progressive normalization of bioelectrical, biochemical, and biomechanical parameters. Monitoring these markers during healing may reveal gradual restoration of neuromuscular coordination, reduction of inflammatory signals, recovery of muscle oxygen utilization efficiency, and stabilization of mechanical properties such as stiffness and strength[70]. Longitudinal multimodal tracking enables quantification of recovery trajectories, where coordinated normalization across neuromuscular, mechanical, and metabolic domains over intermediate time scales indicates progression from injury toward functional recovery.
Therefore, rather than relying on single-parameter thresholds, current evidence supports a potentially feasible, pattern-based interpretation framework combining bioelectrical, biochemical, mechanical, and temporal features to infer transitions among fatigue, injury, and healing states[71-73] [Table 1].
Multimodal physiological characteristics and temporal features of muscle fatigue, injury and recovery
| Muscle health state | Primary biological nature | Main changing indicators | Temporal scale | Reversibility |
| Fatigue | Functional decline (without structural disruption) | MF ↓, RMS ↑, lactic acid ↑, muscle stiffness ↑, muscle strength ↓ | Short-term (minutes to hours) | Rapid recovery (after rest) |
| Injury | Structural disruption (myofibers and extracellular matrix) | CK ↑, IL-6 ↑, muscle stiffness ↑, muscle strength ↓ | Intermediate to long-term (days to weeks) | Limited or delayed recovery (without intervention) |
| Recovery | Structural remodeling and functional restoration | Toward baseline | Intermediate to long-term (days to weeks) | Progressive restoration toward baseline |
WEARABLE SENSORS FOR MUSCLE HEALTH MONITORING
Wearable sensors have become a promising route for realizing continuous, real-time and non-invasive monitoring of muscle functional status in the field of sports and rehabilitation[56,57] [Table 2]. To enable meaningful interpretation of wearable-derived signals in muscle health monitoring, it is essential to recognize that individual physiological markers rarely provide definitive diagnostic information in isolation. Instead, wearable sensing technologies should be applied within a multimodal framework that integrates neuromuscular activity, biomechanical performance, metabolic stress, and recovery trajectories over time. In this context, wearable systems are particularly valuable for identifying functional deviations from individualized baselines, monitoring dynamic transitions across fatigue, injury risk, and recovery phases, and supporting longitudinal assessment in real-world athletic and daily exercise environments.
Sensing technologies for muscle health monitoring
| Sensor type | Target signal | Detection principle | Sensitivity | Response time | Detection range | Limit of detection | Technological maturity | Main advantages | Current limitations | Comfort | References | |
| Bioelectrical sensors | sEMG | Bioelectrical signal acquisition | NA | NA | NA | NA | High | Real-time analysis; High temporal resolution; Non-invasiveness | Electrode placement sensitivity; Motion artifacts; Cross-talk | Medium | [74] | |
| Biochemical sensors | Energy metabolism biomarkers | Lactic acid | Enzyme-based electrochemical sensing | ~ 644.2 nA/mM | 1 min | 0-30 mM | 1 mM | Medium | Real-time analysis; Non-invasiveness; Ease to use | Low integration; Lab validation only | High | [75] |
| Colorimetry | ~ 12.3 /decade | NA | 0-100 mM | 1.5 mM | High | Real-time analysis; Ease to use | Fabrication complexity | High | [76] | |||
| Urea | Enzyme-based electrochemical sensing | ~ 60 mV/decade | 5 min | 0-200 mM | 5 mM | Low | Ease to use | Lab validation only | Medium | [77] | ||
| Colorimetry | ~ 30 /decade | 4 min | 0-30 mM | 5 mM | Low | Multiplex detection | Lab validation only | Medium | [78] | |||
| Muscle injury biomarkers | Mb | Immunochromatography | ~ 0.001 /(ng/mL) | 5 min | 0-1000 ng/mL | 0.05 ng/mL | Low | Multiplex detection; High sensitivity | Specialized equipment dependence; Lab validation only | Low | [79] | |
| Electrochemical transduction | ~ 0.023 V/decade | 1 min | 0-1 × 104 ng/mL | 0.011 ng/mL | Low | Self-powered; Ultra-sensitivity | Architecture complexity; Lab validation only | Low | [80] | |||
| CK | Immunochromatography | ~ 0.014 /(ng/mL) | 5 min | 0-250 ng/mL | 0.063 ng/mL | Medium | Multiplex detection; High sensitivity | Specialized equipment dependence; Lab validation only | Low | [79] | ||
| Enzyme-based electrochemical glucose sensing | ~ 0.0147 /(U/mL) | 5 min | 0-0.5 U/mL | 0.00147 U/mL | Low | Label-free; Ease to use | Cascade reaction dependent; Indirect detection; Lab validation only | NA | [81] | |||
| Inflammatory biomarkers | IL-6 | Aptamer-based electrochemical sensor | ~ 0.62 /(ng/mL) | 1 min | 0-30 ng/mL | 1.3 ng/mL | Medium | Multiplex detection; Clinical validation | Fabrication complexity | Medium | [82] | |
| Colorimetry | ~ 0.006 /(μg/mL) | 5 min | 0-125 μg/mL | 1.95 μg/mL | Low | Label-free; Simple readout | Narrow range; Lab validation only | NA | [83] | |||
| TGF-β | Aptamer-based electrochemical sensor | ~ 0.12 /(pg/mL) | 1 min | 0-150 pg/mL | 6.0 pg/mL | Medium | Multiplex detection; Clinical validation | Fabrication complexity | Medium | [82] | ||
| Optical immunoassay | ~ 0.08 nm/decade | 20 min | 0-108 pg/mL | 100 pg/mL | Low | Ultra-sensitivity; Intracellular detection; Real-time | Lab validation only | NA | [82] | |||
| Biomechanical sensors | Muscle strength | Bioelectrical signal acquisition | ~ 0.0061 mV/kg | 2 s | 0-20 kg | 2.5 kg | Medium | Low impedance; Wireless | Design complexity; Lab validation only | High | [84] | |
| Piezoelectricity (Strain) | ~ 0.066 V/% | NA | -30%-30% | NA | Medium | Ultra-compliance; High repeatability | Lab validation only | High | [85] | |||
| Tissue stiffness | Micro electro mechanical systems | ~ 0.04 μV/kPa | 1 min | 0-1600 kPa | 25 kPa | Medium | Ease to use; Clinical validation | Design complexity | Medium | [86] | ||
| Piezoelectricity | ~ 0.125 μV/kPa | 1 min | 0-160 kPa | 8 kPa | Medium | Small-lesion; Real-time; Clinical validation | Fabrication complexity | Low | [87] | |||
| Elastography | ~ 0.085 /(m·kPa) | 0.6 ms | 0-1532 kPa | 88 kPa | Medium | Depth-sensitivity; Motion-tolerance | Lab validation only | High | [88] | |||
| Near-infrared spectroscopy | ~ 0.35 Hz/kPa | 0.3 s | 0-800 kPa | 20 kPa | Medium | High spatial resolution | Fabrication complexity; Lab validation only | High | [89] | |||
Wearable bioelectrical sensors
sEMG enables recording of the electrical activity of skeletal muscles driven under neural drive. Owing to its non-invasive nature and wide applicability, it has emerged as a key technology for assessing exercise-related muscle fatigue[34,90]. Implementation of wearable technology: Typically, fabric electrodes or array electrodes are adopted, with skin-electrode contact optimized via conductive fibers and gels. Combined with low-noise amplification, anti-power frequency and motion pseudo-aberration circuits, the signal front-end conditioning is achieved. Feature extraction and pattern recognition in the time-frequency domain are enabled through embedded or edge computing[84,91]. High-density sleeve arrays further break through the limitations of complex muscle group monitoring, enabling motion recognition and continuous joint angle estimation in areas such as the hand, demonstrating feasibility and low latency advantages in human-computer interaction and rehabilitation scenarios[92,93].Compared to traditional single-electrode arrangements, arrayed wearables mitigate performance fluctuations caused by electrode placement dependence and drift[94]. However, sweat interference, conductive media drying, and fabric slippage during dynamic movements still affect signal-to-noise ratio and reproducibility, necessitating resolution through integrated structural design and domain adaptive algorithms. Currently, sEMG has been widely applied in training and rehabilitation fields, including muscle group recruitment pattern recognition, posture/movement phase annotation, and intent decoding interfaces coordinated with external biomechanical quantities, providing technical support for individualized training prompts and rehabilitation assistive device control[95-97].
ECG is primarily used to evaluate cardiac rhythm and conduction function[98-100]. By analyzing the adjacent R-R intervals, indicators of heart rate (HR) and heart rate variability (HRV) can be derived; both serve as quantitative parameters for characterizing overall cardiopulmonary load and indirectly reflecting the impact of exercise on the body’s overall physiological state[101]. During exercise, ECG signals are prone to interference from exercise artifacts, leading to reduced accuracy of the indicators[102,103]. Meanwhile, overall physiological indicators like HR and HRV cannot accurately reflect the local fatigue or repair status of specific muscle groups. Therefore, they are more suitable for joint interpretation with local physiological signals such as sEMG. Through multi-dimensional data fusion of “overall + local”, the accuracy of muscle function assessment can be improved[104].
Nevertheless, sEMG and ECG signals are susceptible to electrode placement variability, cross-talk, and motion artifacts, which may affect reproducibility and quantitative consistency across individuals and during dynamic activities. Thus, current bioelectrical wearables are better suited for relative, qualitative monitoring rather than absolute, quantitative measurement.
Wearable biochemical sensors
The balance between energy substrate consumption and synthesis, along with the level of inflammatory response, constitutes a key dimension for assessing the extent of muscle fatigue, damage, and repair efficiency[105]. Core biomarkers reflecting this balance encompass reserved glycogen, lactate, LDH, UN, Mb, CK and IL-6. These biomarkers are present in blood, sweat, and interstitial fluid, with their concentration dynamics exhibiting significant cross-correlations[106,107]. Therefore, measurements obtained from sweat or interstitial fluid can be utilized to estimate their corresponding levels in blood, thereby providing a non‑invasive or minimally invasive analytical approach for evaluating muscle metabolic status.
For energy metabolism monitoring
The combined monitoring of lactic acid and LDH offers complementary biological clues for evaluating muscle fatigue injury, spanning the dual dimensions of “metabolic intensity-structural damage”[108]. Together, these biomarkers exhibit clear synergistic value in the muscle physiological function chain.
As a key metabolite in anaerobic glycolytic, lactic acid concentration directly reflects the intensity of muscle metabolic load and is technically amenable to continuous in vivo monitoring. It stands out as one of the most clinically translatable wearable biochemical markers to date. By integrating the lactic acid oxidase catalytic system with an amperometric signal readout module, researchers can precisely measure lactic acid levels. Meanwhile, the microfluidic structure embedded within wearable patches stabilizes sweat flux and mitigating interference from sweat rate fluctuations[109]. Importantly, such patches can be integrated with electrophysiological detection channels (e.g., ECG), and low-power wireless transmission module, enabling synchronous multi-signal acquisition and transmission across the “metabolism-electrophysiology”[110]. Technical prototypes have demonstrated dynamic monitoring and wireless transmission of lactic acid concentration in real scenarios, including sports training and high-intensity interval training, fully verifying their potential in sports and rehabilitation applications[109]. In contrast, LDH - a key enzyme regulating the conversion of lactic acid to pyruvate - indirectly indicates muscle cell membranes integrity through activity changes (e.g., enzyme release caused by injury), positioning it as a potential marker of muscle structural damage[111]. However, wearable detection of protein markers such as LDH remains hindered by technical bottlenecks, limiting its development to the methodological exploration stage without reliable real-world applications[112,113].
Sweat urea and blood urea, originating from distinct body fluid chambers differ fundamentally in their physiological information and cannot be directly equated or interchanged[17]. They occupy unique niches in muscle training and rehabilitation monitoring. As an end product of nitrogen metabolism, sweat urea provides dynamic insights into individualized metabolic status (e.g., protein breakdown intensity) and hydration levels during training and rehabilitation[111], making it a promising target for wearable monitoring. Sweat urea detection relies on urease as the core biometric recognition element, converting urea concentration into measurable signals via specific catalytic reactions, with common detection principles including potentiometric method or amperometric method[114,115]. To support long-term monitoring, devices typically incorporate microfluidic flow-limiting structure (to stabilize the flow of sweat samples), skin adhesives optimized for sweaty regions (to enhance sample collection efficiency), and a low-power wireless transmission module (for real-time data feedback), ensuring continuous recording during dynamic scenarios such as training and recovery[116]. Emphasizing “trend tracking” (e.g., urea concentration fluctuations across exercise cycles) over precise quantification, sweat urea monitoring provides scenario-based guidance for metabolic load adjustment and hydration optimization[109].
Conversely, blood urea detection via interstitial fluid (ISF) sampling using microneedle arrays, combined with electrochemical or colorimetric immunoassays, shows certain potential due to the correlation between ISF and blood urea concentrations[117]. However, this scheme still faces unresolved challenges: skin safety concerns with microneedle puncture, device stability during prolonged wear (e.g., microneedle blockage, signal drift), and interindividual calibration of ISF-to-blood urea concentration. These hurdles delay its clinical-grade application in the near term[118].
For muscle injury monitoring
Mb and CK can form an integrated indicator pair for “acute injury-progression assessment”: Mb rises within hours after loading and peaks earlier than CK, making it suitable for identifying early intervention timing post-exercise[119,120]. CK rises at 12-24 h and peaks at 24-72 h, positioning it for evaluating decision points in load progressive/ return-to-activity protocols[121].At present, detection relies primarily on spot testing of serum or fingertip/urine samples; non-invasive, continuous wearable monitoring remains immature. A more realistic approach involves using microneedle patches to collect interstitial fluid, combined with immunoelectrochemical or coupled enzyme activity colorimetry, as a phased retesting tool during post-match and recovery periods. Within the continuous training-competition-recovery[122], Mb for early screening within hours and CK for progression assessment within 24-72 hours[47] are recommended, alongside joint interpretation with continuous signals like sEMG[123]. Decisions on load adjustment and return to exercise should be based on individual baselines and relative changes. Limitations such as minimal invasiveness, time lag and individual differences require attention. Overall, this approach is best positioned as research/quasi-implementation, serving as a structural supplement to continuous signals[124].
For inflammatory regulation monitoring
During the entire cycle of muscle fatigue injury and repair, inflammation-related factors, such as Interleukin-6 (IL-6), and transforming growth factor-β (TGF-β), exhibit phased dynamic changes along the temporal axis of “injury initiation-inflammatory response-tissue remodeling”. These temporal patterns provide critical biological insights for evaluating muscle inflammatory states and determining repair processes at the molecular level[125]. Among them, IL-6, a prototypical pro-inflammatory cytokine, serves as an indicator of early post-injury inflammatory activation, while TGF-β modulates tissue remodeling during the middle stage phase. Together, these cytokines form key molecular targets for monitoring the inflammation-repair process. Current wearable technologies for detecting inflammatory factors utilize ISF as the primary sample[118,126]. Through electrochemical (e.g., amperometric, potentiometric) or optical (e.g., fluorescence quenching, colorimetric) techniques, these devices convert biomolecular binding events - between inflammatory factors and their recognition elements - into quantifiable electrical or optical signals. This approach enables highly selective capture of inflammatory factors such as IL-6 and TGF-β, minimizing interference from other body fluids constituents[127].
Wearable monitoring of inflammatory factors remains in the stage of application expansion and experimental verification, yet to reach practical implementation. Overcoming technical bottlenecks would enable it to complement existing metabolic dimension (e.g., sweat lactic acid) and mechanical (e.g., muscle strength and stiffness) indicators already in preliminary use, addressing the gap in molecular-level monitoring of muscle inflammatory states[128]. This advancement would further strengthen the evidence chain for comprehensive assessment of muscle fatigue, injury, and repair cycles, offering a more robust molecular foundation for precise interventions - such as optimizing anti-inflammatory timing and refining rehabilitation protocols.
Despite the progress achieved so far, wearable biochemical sensing still faces inherent challenges. Analyte levels in sweat or interstitial fluid may not directly reflect blood concentrations due to compartmental differences and temporal delays, while low biomarker abundance increases susceptibility to noise and cross-reactivity. Signal stability can also be affected by sweat variability, biofouling, and calibration drift, particularly during dynamic exercise. Therefore, current wearable biochemical sensors are better suited for trend monitoring rather than definitive diagnostic use.
Wearable biomechanical sensors
For muscle strength and dynamic kinematics assessment
The direct measurement of narrow “muscle strength” - such as isokinetic and isometric contraction force - relies on gold standards instruments including isokinetic force gauges, isometric force gauges and handgrip force gauges[129,130]. These tools offer high objectivity and repeatability while providing core metrics like peak muscles contraction force[131]. For dynamic tasks involving complex movements (e.g., running, jumping and throwing), mechanical analysis requires integration of marker-based three-dimensional optical motion capture systems with ground force platforms. By collecting kinematic data (e.g., joint angles and displacements) and external force data (e.g., ground reaction forces), inverse dynamics algorithms enable computation of net joint torques and other mechanical outputs, facilitating detailed biomechanical profiling of dynamic movements. However, such laboratory paradigms are limited by high costs and dependence on controlled scenarios (e.g., specialized research facilities), restricting their utility primarily to benchmarking the accuracy of wearable-based mechanical estimation methods. In contrast, inertial measurement unit (IMU) and smart insoles have emerged as core devices for monitoring muscle strength-related indicators in dynamic scenes[22]. Their integration enables inverse estimation of three-dimensional ground reaction force (GRF), plantar pressure center, and derived metrics such as gait event timing and vertical stiffness (Kvert)[132]. These indicators directly inform assessments of muscle fatigue (e.g., abnormal decline in vertical stiffness indicating lower limb muscle fatigue) and recovery trajectories (e.g., symmetrical recovery of GRF reflecting improved muscle strength balance)[133]. Critically, these wearable systems operate independently of controlled environments, supporting long-term continuous monitoring in naturalistic settings such as training grounds and rehabilitation centers - thus addressing the contextual limitations of traditional laboratory techniques.
For soft tissue stiffness evaluation
Traditional laboratories employ ultrasonic shear wave elastography or strain imaging techniques. By emitting shear waves into tissues and analyzing their propagation speeds, the equivalent modulus of the tissues is quantified, achieving high measurement accuracy. However, this technology relies on large-scale equipment, requires professional operation, and is restricted to static or controlled tasks - such as measuring muscle contraction in fixed posture - failing to meet the demand for continuous recording in motion scenarios[134]. Consequently, its adaptability across diverse settings remains limited. In recent years, wearable elastography technology based on the mechanism of “electromechanical vibration-skin surface wave dispersion analysis” has demonstrated significant advantages: wireless transmission, low power consumption, and centimeter-level depth resolution. Importantly, its measurement results have been verified to align with those of ultrasonic elastography[135]. This innovation enables continuous tracking of muscle stiffness during dynamic activities, including walking and resistance training. For example, it detects localized stiffness increase post-injury and gradual stiffness decrease during recovery. Nevertheless, further optimization is still required to enhance dynamic accuracy and long-term stability.
Overall, wearable biomechanical sensors enable practical estimation of soft tissue stiffness in real-world settings. However, their outputs are derived from indirect modeling rather than direct force measurement, introducing uncertainty during highly dynamic activities. Sensor misalignment, soft tissue artifacts, and biomechanical assumptions may further affect accuracy. Therefore, current wearable systems should be considered complementary tools rather than replacements for gold-standard laboratory instrumentation.
It deserves to note that, in practical applications, wearable sensors do not typically diagnose muscle fatigue, injury, or healing as discrete clinical labels. Instead, they quantify deviations from individualized physiological baselines and monitor temporal patterns that may indicate transitions between functional states. In competitive sports, sustained reductions in neuromuscular efficiency and abnormal recovery trajectories may serve as early warning signs of injury risk following cumulative fatigue. In public physical exercise settings, wearable systems can detect excessive training loads by identifying persistent elevations in muscle activation relative to output performance. During rehabilitation and healing, progressive normalization of mechanical and neuromuscular indicators may provide objective metrics for recovery monitoring and safe return-to-activity decision-making. Thus, wearable technologies currently function primarily as continuous monitoring and risk assessment tools rather than standalone diagnostic instruments.
APPLICATIONS OF WEARABLE MUSCLE HEALTH MONITORING IN REAL-WORLD SETTINGS
Exercise intensity and training load monitoring
Precise monitoring of exercise intensity and training load is a core link in optimizing training plans, preventing sports injuries, and enhancing sports performance. It is also a research hotspot and practical focus in the field of sports science. Traditional load monitoring mostly relies on macro indicators such as external training volume (such as running volume, number of sets and number of repetitions), which is difficult to accurately reflect the molecule-level physiological response of athletes and the stress state of their muscles because of monitoring lag and insufficient data dimensionality. With the rapid development of wearable sensing technology, biomechanical analysis and artificial intelligence algorithms, a real-time monitoring system based on multi-dimensional physiological and mechanical signals has emerged. Such a system can achieve refined and individualized assessment of exercise intensity and training load based on detailed analyses on neuromuscular function, mechanical load and cardiopulmonary metabolism.
Strength monitoring through sEMG
Load monitoring can be carried out through muscle electrical signals to reflect the activation mode of muscle groups and the degree of force application, which is the core physiological basis for evaluating exercise intensity. sEMG and high-density EMG (HD-EMG) technologies, with their non-invasive and real-time advantages, have become key means to capture neuromuscular activity signals. The form and function of their equipment are constantly upgrading towards flexibility, high precision, and multi-dimensionality.
For instance, the clothing-grade sEMG technology integrates flexible electrodes into conductive fabric clothing (such as forearm covers, shorts, etc.), and pairs them with miniaturized signal amplification modules and on-board computing units to synchronously collect EMG signals from multiple muscle groups. By analyzing the signal characteristics of target muscle groups under different weight loads, and mapping the internal muscle load to external training volume (like sets, repetitions and speed), a training intensity range that conforms to the individual muscle strength level of athletes can be established for “total load” assessment. It provides precise data support for the dynamic adjustment of training load to avoid long-term overloading of local muscle groups and reduce muscle strain risk[136].
The HD-EMG sleeve integrates up to 150 microelectrodes to achieve high-density and high-resolution EMG collection from the forearm muscle groups. By using machine learning algorithms to extract and analyze the features of EMG signals within a 100 ms time window, > 30 gesture movements can be accurately decoded and continuous joint angle regression prediction can be achieved (prediction error of < 10°), providing a powerful tool for analyzing the neuromuscular control mechanism underlying decomposed technical movements. In events with extremely high requirements for movement accuracy such as throwing, hitting, gymnastics, fencing, and racket sports, HD-EMG can depict the muscle fingerprints of athletes of different levels (from novices to experts) or the same athlete in different states (from normal to injured and rehabilitated). By comparing features such as the recruitment sequence of muscle groups and the distribution of activation intensity, one can quantitatively evaluate technical movements accuracy and rehabilitation training effects, and this lowers the risk of music-tendon system injuries[92].
Mechanical load and run-jump vertical stiffness monitoring
Mechanical load and running and jumping vertical stiffness (resistance of the human body against vertical deformation upon vertical loading, termed as Kvert) characterize the mechanical features and elastic state of the lower limb muscle-tendon system during exercise. Their dynamic changes are closely related to exercise performance, fatigue degree and injury risk. The popularization of wearable devices such as IMUs and smart pressure insoles has enabled on-site and real-time monitoring of key mechanical parameters such as contact time, flight time, and Kvert. The fluctuation pattern of Kvert is believed to correlate with muscle fatigue and the functional state of the muscle-tendon system. Through long-term monitoring of Kvert and gait parameters within training sessions or across the entire season, the leg mechanical load plot for athletes can be constructed to visually reflect the accumulation and recovery status of the load. It is beneficial to offer data support for the modifying the training cycle planning. For example, when Kvert of one leg is persistently lower than that of the other for a long time, or shows an abnormal decrease with the increase of training volume, it often refers to conditions such as muscle fatigue, soft tissue micro-injury or abnormal force line in that lower extremity. In other words, it can be used as an early warning sign for overloading training and potential injury[137,138].
Cardiopulmonary and metabolic intensity monitoring
Cardiopulmonary and metabolic intensity reflect the energy supply status and overall physiological stress level of athletes during exercise, and they are the key basis for formulating endurance training plans and assessing the recovery status of physical fitness. Wearable devices incorporating sweat-based biochemical sensing and photoplethysmography (PPG) have enabled real-time monitoring of core metabolites (e.g., lactic acid, ions) and heart rate, promoting the development of metabolic and cardiopulmonary intensity assessment towards a non-invasive and continuous manner[139]. For example, the chemical-electrophysiological hybrid patch integrates lactase electrodes and ECG electrodes on a flexible substrate, achieving stable collection of sweat lactate concentration and heart rate[140] [Figure 2A]. The patch shows negligible cross-interference for sensing the two biomarkers, empowering simultaneous tracking of physiological responses of the local muscle metabolism and the systemic cardiovascular system. It realizes multi-dimensional comprehensive determination of exercise intensity by overcoming the limitations of traditional analysis based on single indicators. Moreover, for groups such as high-level endurance athletes and rehabilitation trainees with heart disease history who need precise intensity control, this patch ensures the safety of the training process via the hybrid physiological monitoring.
Figure 2. Real-world applications of wearable muscle health monitoring in competitive sports and public physical exercises; (A) Monitoring of exercise intensity and training load based on ECG and lactic acid sensing. Reproduced with permission[140]. Copyright 2016, Springer Nature; (B) Assessment of athlete’s muscle fatigue based on sEMG and muscle strength measurement. Reproduced with permission[142]. Copyright 2025, John Wiley and Sons; (C) Formulation of training recovery based on lactate and urea sensing. Reproduced with permission[145]. Copyright 2022, Springer Nature; (D) Nutrition uptake and metabolic analysis based on AI-empowered multimodal sensing. Reproduced with permission[151]. Copyright 2021, American Chemical Society. W.E.: working electrode; R.E.: reference electrode; C.E.: counter electrode; ECG: electrocardiography; AI: artificial intelligence; sEMG: surface electromyography.
Athletes’ health and fatigue status assessment
The precise assessment of athletes’ health and fatigue status is a vital prerequisite for the scientific training of sports, the pre-emptive prevention of injuries, and the optimization of competitive performance. It is also a hot topic in the field of sports science and sports medicine. The burgeoning of wearable sensing has brought about promising opportunities with theoretical references and technical support for the construction of a scientific and efficient athlete health management and fatigue monitoring system.
Peripheral muscle fatigue assessment
Peripheral muscle fatigue is characterized by the decline in muscle function after exercise. The core mechanism involves the disorder of energy metabolism in muscle fibers, the imbalance of ion homeostasis and the reduction of neuromuscular transmission efficiency. Multi-channel sEMG and HD-EMG techniques, are favorable methods for quantifying peripheral muscle fatigue due to their advantageous non-invasiveness and high spatiotemporal resolution. By comparing the EMG spectral characteristics and spatial distribution patterns before and after training or during multiple training weeks, it is possible to objectively determine potential signs of premature fatigue in the muscles, such as premature spectral decline and hyperactivity of the synergic muscles. This is helpful for the setting of load progression rhythm and rest intervals in the training plan to avoid functional decline caused by overtraining[141]. Moreover, for athletes with a history of muscle injury, in progressive load tests, employing sEMG to monitor the electrophysiological responses of injured muscle groups can identify the abnormal pattern of early fatigue and compensatory activation, therefore assisting in determining the safe load threshold in case of injury.
Soft tissue stiffness and muscle injury assessment
Soft tissue stiffness changes are important biomechanical indicators for muscle injury, fatigue accumulation and healing process. Acute muscle strain is often accompanied by an increase in local tissue stiffness, and the abnormal elasticity of scar tissue in the later stage of injury may affect muscle function recovery and sports performance. Traditional assessment methods employ ultrasonic shear wave elastography and magnetic resonance imaging to accurately measure tissue stiffness. But they are limited by complex operation, high cost, and difficulty in frequent clinical visit, and thus cannot meet the dynamic monitoring requirements in training grounds or daily lives. The emergence of wireless mechano-acoustic wave (MAW) elastography patches has effectively broken through this technical bottleneck with easy wearing, cost effectiveness and the capability of long-term tracking. MAW elastography patches can continuously monitor the stiffness changes of the injured area on the training ground or in daily life, accurately assess the healing stage, and provide an objective basis for load adjustment in rehabilitation training. Especially during the concentrated season and the high-incidence period of injury such as re-loading after injury, long-term monitoring of stiffness dynamics can be conducted on easily injured muscle groups such as the posterior thigh muscles and the triceps surae. When it is found that the muscle stiffness continuously increases, it indicates a great chance of potential re-injury and calls for adjustment of the training plan[142] [Figure 2B].
Formulation of training recovery and nutrition strategies
The scientific formulation of training recovery and nutrition strategies plays an important role in maintaining competitive state, easing fatigue and preventing injuries. It is also the core direction for the transformation of the field of sports science from “empirical regulation” to “precise intervention”. Emerging wearable sensing technology can capture key physiological signals in the recovery process of athletes in real time and in a multimodal way, such as metabolite levels, fluid balance status, body composition changes and tissue repair microenvironment. The systemic analysis of these markers is propelling the transformation of sports science from empirical regulation to precise intervention.
Hydration status of the body and electrolyte management
Sweat is an important medium for the excretion of metabolites and electrolytes during exercise. The dynamic changes in its components (such as lactic acid, glucose, electrolytes, urea, etc.)[40], secretion rate and total amount are associated with the metabolic load, hydration status and physiological stress level of athletes. Wearable sweat sensing technology integrates multiple types of sensors to achieve real-time and non-invasive monitoring of multiple biochemical indicators (like glucose, lactic acid, Na+ and K+). It accurately captures the physiological responses under different hydration states, as well as the dynamic change patterns of Na+/K+ concentrations under hydrated and non-hydrated conditions. The technology enables accurate assessment of the dehydration risk and electrolyte loss characteristics for professional athletes, and therefore aids the formulation of targeted fluid replacement volume, timing and electrolyte supplementation plans to effectively reduce the risk of heat-related diseases such as muscle spasms and heat exhaustion[13]. The battery-free, short-range wireless communication microfluidic patch further expands the monitoring modes via integrating electrochemical detection (lactic acid, glucose), colorimetric analysis (pH, Cl-), and volumetric measurement (sweat rate, total sweat volume) within a single device. Long-term tests reveal an explainable time-lag correlation between its sweat-based detection results and corresponding marker levels in blood. Employing the patch may offer objective clues for reasonably adjusting the interval time, training intensity and recovery interval, by recording the lactate-power curve and tracking the metabolic stress distribution and recovery process of interval training[143,144] [Figure 2C].
Regulation of the microenvironment for tissue repair
The quality of tissue repair after local muscle-fascia injury directly affects the recovery process and re-injury risk of athletes. Traditional repair strategies mostly focus on mechanical rehabilitation training and lack precise regulation of the local microenvironment of the injured tissue. The development of mechanically active therapy provides a new route for precise tissue repair: by programming the local stress/strain state to optimize the mechanical microenvironment of the injured region, it promotes the healing of injured tissues[145,146]. It is anticipated to combine such active tissue mechano-modulation with wearable stiffness sensing to establish monitoring-intervention closed-loop injury repair strategy. Specifically, the healing status of injured muscle or tendon can be evaluated by wearable stiffness monitoring, and this in turn, based on biomechanical and mechanobiological pro-healing mechanisms, guides the rational design of mechanical loading parameters to accelerate healing and avoid scar formation[147,147].
AI-enabled multimodal tracking
Unimodal fatigue monitoring technologies - particularly those based on sEMG - have long dominated the field. However, increasing empirical evidence suggests that reliance on a single physiological signal is insufficient for achieving stable and reliable muscle fatigue assessment. The limitations primarily include high sensitivity to inter-individual variability, limited capability in identifying transitional states, and susceptibility to motion artifacts during dynamic exercise.
Combined with artificial intelligence (AI) algorithms, unimodal fatigue evaluation based on wearable sEMG sensing shows reasonable average recognition performance, however with substantial inter-subject variability[35]. Specifically, sEMG coupled with support vector machine (SVM) achieves peak accuracy over 90%, yet the lowest individual accuracy is merely < 65%. Compared to unimodal sEMG with temporal convolutional network (TCN) processing, a dual-modal TCN model integrating ECG and sEMG exhibits significantly improved fatigue classification performance, with an overall average accuracy of 88.5% and an inter-personal variation range of ~ 10% (> 20% for unimodal model). In another study, a multimodal framework integrating muscle thickness, joint angle, and sEMG is developed to reduce the false positive rate down to 3%, thereby enhancing the functional robustness under dynamic conditions and improving reliability in real-world training scenarios[148]. Expanding multimodal frameworks to biochemical sensing, the coordinated analysis of metabolism-related biochemical markers (i.e., sweat glucose and lactate) and sEMG also aids partial differentiation of fatigue levels[149]. These findings highlight that the reliability of wearable fatigue detection can be solidified by integrated evaluations of neuromuscular activation, mechanical behavior, cardiovascular regulation, and metabolite accumulation. In this context, multimodal data fusion combined with AI analysis extends fatigue assessment beyond state recognition toward decision support.
The increasingly reliable fatigue evaluation would also benefit quantitative training load regulation and recovery management. Since muscle fatigue is regarded as the cumulative physiological stress responding to external training stimuli, lower estimation error of training load can be realized with refined evaluation of fatigue level, leading to more physiologically grounded adjustments of training intensity and volume[148-150]. Furthermore, AI-based analysis of multimodal data, including biomechanical (muscle stiffness and strength) and biochemical (lactic acid, urea and IL-6) indexes, can support objective identification of post-exercise recovery windows. It can also assist in formulating personalized nutritional strategies, such as optimizing the timing and quantity of carbohydrate and protein intake to facilitate energy replenishment and metabolic repair[151] [Figure 2D].
It is worth emphasizing that such performance improvements rely on the capability of AI algorithms to model the complex relationships embedded in multivariate time-series data. In current studies on wearable multimodal sensing, commonly used algorithms include SVM, TCN, bidirectional long short-term memory (Bi-LSTM), and decision-level fusion methods based on Dempster-Shafer (D-S) evidence theory. SVM demonstrates strong generalization performance in small-sample classification scenarios[152-154]. TCN can effectively capture long-term temporal dependencies through dilated convolutions and have outperformed conventional SVM in multimodal fusion models. Bi-LSTM is well suited for handling temporal dependency features and signal drift. Meanwhile, decision-level fusion based on D-S evidence theory further integrates probabilistic information from multiple sources, thereby reducing the influence of inter-individual variability[104].
CONCLUSION AND OUTLOOK
This article systematically elaborates the underlying physiological mechanisms and corresponding bioelectrical, biochemical and biomechanical indicators of muscle fatigue, injury and healing. Based on these, the design and working principle of wearable sensors, as well as their applications in real-world settings for competitive sports and public fitness, including load monitoring, fatigue evaluation and personalized training and nutrition management, are comprehensively introduced.
In spite of the progress made so far, the development of wearable sports monitoring still faces many technical bottlenecks and application challenges. Future research is anticipated to focus on the following aspects [Figure 3]. (1) Technology optimization. Technical breakthroughs overcoming existing bottlenecks such as the stable acquisition of bioelectrical signals (sEMG/HD-EMG) in dynamic environments[155], the long-term monitoring of traced biomarkers (such as CK, IL-6)[17], and the precise estimation of biomechanical parameters (such as ground reaction force) based on IMU and elastography, are demanded. For instance, muscle healing represents a longer-term physiological process that may extend from days to weeks, whereas many current wearable systems are predominantly applied in acute or session-based monitoring scenarios. Continuous or longitudinal tracking of recovery trajectories presents additional challenges, including long-term signal stability, sensor adhesion and biocompatibility, power management, data consistency, and individualized baseline recalibration. Future development of energy-efficient flexible electronics, cloud-based analytics, and personalized modeling strategies will be essential for enabling reliable long-term wearable monitoring of muscle healing. (2) Data fusion. So far, no universally accepted thresholds currently exist that allow wearable signals alone to distinguish muscle fatigue, injury and recovery with diagnostic certainty. Moreover, physiological responses exhibit substantial inter-individual variability, necessitating personalized baseline tracking with fusion of multi-modal signals for refined quantification of muscle function[13]. On this basis, individual characteristics can be incorporated to dynamically and precisely quantify the fatigue, injury and healing status[156]. (3) Closed-loop management. A closed-loop system integrating monitoring, assessment and intervention functions has long been expected to realize personalized and precise sports management. This can be realized through integrating wearable sensing with data-driven injury early warning model depicting the relationship between biomarker level and health status, and intelligent and controllable intervention such as programmable mechanical loading[110]. (4) Interdisciplinary collaboration. Theoretical innovation from molecular mechanisms to wearable technology stems from the in-depth intersection of sports science, biomedicine, engineering and information technology. As for practical applications, it is expected to build an integrated transformation system from lab to the field, by validating and optimizing cutting-edge technological outcomes through large-scale trials, establishing standard norms, and at the same time improving the data security protection[17].
DECLARATIONS
Authors’ contributions
Manuscript structure design: Chang, N.; Su, L.; Liu, H.
Literature review, writing - original draft & revision: Chang, N.; Wu, D.; Song, Y.; Jin, B.
Writing - review & editing: Tang, D.; Yao, T.; Liu, H.
Supervision, project administration: Liu, H.
All authors have read the manuscript and approved the final version.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the grants from the National Natural Science Foundation of China (12302424), the Key Research and Development Program of Shaanxi Province (2023-YBSF-172), the Innovation Chain of Key Industries of Shaanxi Province (2024SF-ZDCYL-01-14), the Key Natural Science Research Project of the Education Department of Anhui Province (K2021ZD0150), the Innovation Team of Xi’an Jiaotong University (xtr062023002), and the Key Scientific Research Project of Xi’an Physical Education University (2025ZD001).
Conflicts of interest
All authors have declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Smith, J. A. B.; Murach, K. A.; Dyar, K. A.; Zierath, J. R. Exercise metabolism and adaptation in skeletal muscle. Nat. Rev. Mol. Cell Biol. 2023, 24, 607-32.
2. Egan, B.; Sharples, A. P. Molecular responses to acute exercise and their relevance for adaptations in skeletal muscle to exercise training. Physiol. Rev. 2023, 103, 2057-170.
3. Gabbett, T. J. The training-injury prevention paradox: should athletes be training smarter and harder? Br. J. Sports Med. 2016, 50, 273-80.
4. Cheung, K.; Hume, P.; Maxwell, L. Delayed onset muscle soreness: treatment strategies and performance factors. Sports Med. 2003, 33, 145-64.
5. Valle, X.; Alentorn-Geli, E.; Tol, J. L.; et al. Muscle injuries in sports: a new evidence-informed and expert consensus-based classification with clinical application. Sports Med. 2017, 47, 1241-53.
6. Edouard, P.; Reurink, G.; Mackey, A. L.; et al. Traumatic muscle injury. Nat. Rev. Dis. Primers 2023, 9, 56.
7. Impellizzeri, F. M.; Shrier, I.; McLaren, S. J.; et al. Understanding training load as exposure and dose. Sports Med. 2023, 53, 1667-79.
8. Martinez-Valdes, E.; Negro, F.; Laine, C. M.; Falla, D.; Mayer, F.; Farina, D. Tracking motor units longitudinally across experimental sessions with high-density surface electromyography. J. Physiol. 2017, 595, 1479-96.
9. Koh, A.; Kang, D.; Xue, Y.; et al. A soft, wearable microfluidic device for the capture, storage, and colorimetric sensing of sweat. Sci. Transl. Med. 2016, 8, 366ra165.
10. Lee, S. P.; Ha, G.; Wright, D. E.; et al. Highly flexible, wearable, and disposable cardiac biosensors for remote and ambulatory monitoring. NPJ Digit. Med. 2018, 1, 2.
11. Wang, R.; Shen, Y.; Qian, D.; et al. Tensile and torsional elastomer fiber artificial muscle by entropic elasticity with thermo-piezoresistive sensing of strain and rotation by a single electric signal. Mater. Horiz. 2020, 7, 3305-15.
12. Li Y, ; Li W, ; Sun A, ; et al. A self-reinforcing and self-healing elastomer with high strength, unprecedented toughness and room-temperature reparability. Mater. Horiz. 2021, 8, 267-75.
13. Li, C.; Wang, H.; Song, Z.; et al. Wireless, wearable elastography via mechano-acoustic wave sensing for ambulatory monitoring of tissue stiffness. Sci. Adv. 2025, 11, eady0534.
14. Cudejko, T.; Button, K.; Al-Amri, M. Wireless pressure insoles for measuring ground reaction forces and trajectories of the centre of pressure during functional activities. Sci. Rep. 2023, 13, 14946.
15. Scheltinga, B. L.; Kok, J. N.; Buurke, J. H.; Reenalda, J. Estimating 3D ground reaction forces in running using three inertial measurement units. Front. Sports Act. Living 2023, 5, 1176466.
16. Ai, Y.; Wang, Z.; Liu, Y.; et al. Robust fiber strain sensor by designing coaxial coiling structure with mutual inductance effect. Adv. Fiber Mater. 2024, 6, 1629-39.
17. Davis, N.; Heikenfeld, J.; Milla, C.; Javey, A. The challenges and promise of sweat sensing. Nat. Biotechnol. 2024, 42, 860-71.
18. Childs, A.; Mayol, B.; Lasalde-Ramírez, J. A.; Song, Y.; Sempionatto, J. R.; Gao, W. Diving into sweat: advances, challenges, and future directions in wearable sweat sensing. ACS Nano 2024, 18, 24605-16.
19. Liu, K.; Meng, Z.; Fang, Y.; Jiang, H. Conductive MOFs for electrocatalysis and electrochemical sensor. eScience 2023, 3, 100133.
20. Yang, Y.; Gao, W. Wearable and flexible electronics for continuous molecular monitoring. Chem. Soc. Rev. 2019, 48, 1465-91.
21. Sempionatto, J. R.; Lasalde-Ramírez, J. A.; Mahato, K.; Wang, J.; Gao, W. Wearable chemical sensors for biomarker discovery in the omics era. Nat. Rev. Chem. 2022, 6, 899-915.
22. Mohamed Refai, M. I.; van Beijnum, B. F.; Buurke, J. H.; Veltink, P. H. Portable gait lab: estimating over-ground 3d ground reaction forces using only a pelvis IMU. Sensors (Basel). 2020, 20, 6363.
23. Baker, L. B.; Model, J. B.; Barnes, K. A.; et al. Skin-interfaced microfluidic system with personalized sweating rate and sweat chloride analytics for sports science applications. Sci. Adv. 2020, 6, eabe3929.
24. Schwiete, C.; Roth, C.; Mester, J.; Broich, H.; Behringer, M. Overlaps of skeletal muscle fatigue and skeletal muscle damage: the muscle injury continuum. Sports Med. Open 2025, 11, 73.
25. Chazaud, B. Inflammation during skeletal muscle regeneration and tissue remodeling: application to exercise-induced muscle damage management. Immunol. Cell Biol. 2016, 94, 140-5.
26. Bestwick-Stevenson, T.; Toone, R.; Neupert, E.; Edwards, K.; Kluzek, S. Assessment of fatigue and recovery in sport: narrative review. Int. J. Sports Med. 2022, 43, 1151-62.
27. Jorgenson, K. W.; Phillips, S. M.; Hornberger, T. A. Identifying the structural adaptations that drive the mechanical load-induced growth of skeletal muscle: a scoping review. Cells 2020, 9, 1658.
28. Paluch, Ł.; Nawrocka-Laskus, E.; Wieczorek, J.; Mruk, B.; Frel, M.; Walecki, J. Use of ultrasound elastography in the assessment of the musculoskeletal system. Pol. J. Radiol. 2016, 81, 240-6.
29. Lee, S. R.; Directo, D.; Kang, Y.; et al. Effects of acute fish oil supplementation on muscle function and soreness after eccentric contraction-induced muscle damage. Nutrients 2025, 17, 3408.
30. Presland, J. D.; Timmins, R. G.; Maniar, N.; et al. Muscle activity and activation in previously strain-injured lower limbs: a systematic review. Sports Med. 2021, 51, 2311-27.
31. Schwartz, F. P.; Celes, R. S.; Bottaro, M.; Nascimento, F. A. Isokinetic work-to-surface electromyographic signal energy ratios as a muscular fatigue indicator. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. 2009, 2009, 1310-3.
32. Sbriccoli, P.; Felici, F.; Rosponi, A.; et al. Exercise induced muscle damage and recovery assessed by means of linear and non-linear sEMG analysis and ultrasonography. J. Electromyogr. Kinesiol. 2001, 11, 73-83.
33. Cifrek, M.; Medved, V.; Tonković, S.; Ostojić, S. Surface EMG based muscle fatigue evaluation in biomechanics. Clin. Biomech. (Bristol). 2009, 24, 327-40.
34. Sun, J.; Liu, G.; Sun, Y.; Lin, K.; Zhou, Z.; Cai, J. Application of surface electromyography in exercise fatigue: a review. Front. Syst. Neurosci. 2022, 16, 893275.
35. Mu, D.; Li, F.; Yu, L.; Du, C.; Ge, L.; Sun, T. Study on exercise muscle fatigue based on sEMG and ECG data fusion and temporal convolutional network. PLoS ONE 2022, 17, e0276921.
36. Li, D.; Chen, C. Research on exercise fatigue estimation method of Pilates rehabilitation based on ECG and sEMG feature fusion. BMC Med. Inform. Decis. Mak. 2022, 22, 67.
37. Yuan, Y.; Cao, D.; Li, C.; Liu, C. Feature fusion of electrocardiogram and surface electromyography for estimating the fatigue states during lower limb rehabilitation. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi 2020, 37, 1056-64. (in Chinese).
38. Chen, S.; Xu, K.; Yao, X.; et al. Information fusion and multi-classifier system for miner fatigue recognition in plateau environments based on electrocardiography and electromyography signals. Comput. Meth. Prog. Bio 2021, 211, 106451.
39. Wang, Y.; Chen, M.; Gao, Y.; et al. Effect of one-time high load exercise on skeletal muscle injury in rats of different genders: oxidative stress and mitochondrial responses. Acta Cir. Bras. 2022, 37, e370805.
41. Elustondo, P. A.; White, A. E.; Hughes, M. E.; Brebner, K.; Pavlov, E.; Kane, D. A. Physical and functional association of lactate dehydrogenase (LDH) with skeletal muscle mitochondria. J. Biol. Chem. 2013, 288, 25309-17.
42. Papadopoulos, D.; Skopas, V.; Trakas, N.; et al. Serum lactate dehydrogenase and its isoenzymes as predictors of clinical outcomes in acute exacerbation of chronic obstructive pulmonary disease: a retrospective analysis of a hospitalized cohort. Monaldi Arch. Chest Dis. 2023, 94, 2543.
43. Cairns, S. P. Lactic acid and exercise performance: culprit or friend? Sports Med. 2006, 36, 279-91.
44. Haralambie, G.; Berg, A. Serum urea and amino nitrogen changes with exercise duration. Eur. J. Appl. Physiol. 1976, 36, 39-48.
45. Prylutskyy, Y.; Nozdrenko, D.; Omelchuk, O.; et al. Effect of C60 fullerene on muscle injury-induced rhabdomyolysis and associated acute renal failure. Int. J. Nanomedicine 2024, 19, 8043-58.
46. Wang, Y.; Liu, D.; Wang, S.; Li, Y.; Liu, G. IBS008738, a TAZ activator, facilitates muscle repair and inhibits muscle injury in a mouse model of sport-induced injury. Cytotechnology 2025, 77, 2.
47. Brancaccio, P.; Lippi, G.; Maffulli, N. Biochemical markers of muscular damage. Clin. Chem. Lab. Med. 2010, 48, 757-67.
48. Hanu, G.; Thota, S.; Venkat, K.; et al. Comparative study of microsurgical lumbar discectomy and percutaneous endoscopic lumbar discectomy based on clinical outcome and muscle injury markers. J. Krishna Inst. Med. S. 2023, 12, 12-9. https://doaj.org/article/2fc9a4b213594359ab7d0654328c2772.
49. Manfredi, T. G.; Fielding, R. A.; O’Reilly, K. P.; Meredith, C. N.; Lee, H. Y.; Evans, W. J. Plasma creatine kinase activity and exercise-induced muscle damage in older men. Med. Sci. Sports Exerc. 1991, 23, 1028-34.
50. Mougios, V. Reference intervals for serum creatine kinase in athletes. Br. J. Sports Med. 2007, 41, 674-8.
51. Clarkson, P. M.; Kearns, A. K.; Rouzier, P.; Rubin, R.; Thompson, P. D. Serum creatine kinase levels and renal function measures in exertional muscle damage. Med. Sci. Sports Exerc. 2006, 38, 623-7.
52. Abaïdia, A. E.; Lamblin, J.; Delecroix, B.; et al. Recovery from exercise-induced muscle damage: cold-water immersion versus whole-body cryotherapy. Int. J. Sports Physiol. Perform. 2017, 12, 402-9.
53. Martínez-Navarro, I.; Collado, E.; Hernando, C.; Hernando, B.; Hernando, C. Inflammation, muscle damage and postrace physical activity following a mountain ultramarathon. J. Sports Med. Phys. Fitness 2021, 61, 1668-74.
54. Reid, M. B.; Lännergren, J.; Westerblad, H. Respiratory and limb muscle weakness induced by tumor necrosis factor-alpha: involvement of muscle myofilaments. Am. J. Respir. Crit. Care Med. 2002, 166, 479-84.
55. Hardin, B. J.; Campbell, K. S.; Smith, J. D.; et al. TNF-alpha acts via TNFR1 and muscle-derived oxidants to depress myofibrillar force in murine skeletal muscle. J. Appl. Physiol. (1985). 2008, 104, 694-9.
57. Karlsen, A.; Mackey, A. L.; Suetta, C.; Kjaer, M. What is the impact of acute inflammation on muscle performance in geriatric patients? Exp. Gerontol. 2020, 138, 111008.
58. Huang, C.; Ogawa, R. Mechanotransduction in bone repair and regeneration. FASEB J. 2010, 24, 3625-32.
59. Khoury, V.; Guillin, R.; Dhanju, J.; Cardinal, E. Ultrasound of ankle and foot: overuse and sports injuries. Semin. Musculoskelet. Radiol. 2007, 11, 149-61.
60. Guilhem, G.; Doguet, V.; Hauraix, H.; et al. Muscle force loss and soreness subsequent to maximal eccentric contractions depend on the amount of fascicle strain in vivo. Acta Physiol. (Oxf). 2016, 217, 152-63.
61. Elmer, S.; McDaniel, J.; Mattson, J.; Martin, J. Effect of a contusion injury on muscular force, power, work, and fatigue. Scand. J. Med. Sci. Sports 2012, 22, 488-94.
62. Silver, J. S.; Günay, K. A.; Cutler, A. A.; et al. Injury-mediated stiffening persistently activates muscle stem cells through YAP and TAZ mechanotransduction. Sci. Adv. 2021, 7, eabe4501.
63. Lieber, R. L.; Fridén, J. Muscle damage is not a function of muscle force but active muscle strain. J. Appl. Physiol. (1985). 1993, 74, 520-6.
64. Yanagisawa, O.; Sakuma, J.; Kawakami, Y.; Suzuki, K.; Fukubayashi, T. Effect of exercise-induced muscle damage on muscle hardness evaluated by ultrasound real-time tissue elastography. Springerplus 2015, 4, 308.
65. Kharraz, Y.; Guerra, J.; Mann, C. J.; Serrano, A. L.; Muñoz-Cánoves, P. Macrophage plasticity and the role of inflammation in skeletal muscle repair. Mediators Inflamm. 2013, 2013, 491497.
66. Alba-Jiménez, C.; Moreno-Doutres, D.; Peña, J. Trends assessing neuromuscular fatigue in team sports: a narrative review. Sports (Basel). 2022, 10, 33.
67. Pitzalis, R. F., Lagomarsino, B., Ceroni, I., et al. Evaluating muscle fatigue with non-invasive approaches: a review of methods, metrics, and implications. IEEE Trans. Neural Networks Learn. Syst. 2025, 33, 2707-23.
68. Smith, C.; Kruger, M. J.; Smith, R. M.; Myburgh, K. H. The inflammatory response to skeletal muscle injury: illuminating complexities. Sports Med. 2008, 38, 947-69.
69. Jaén-Carrillo, D.; Pérez-Castilla, A.; García-Pinillos, F. Wearable and portable devices in sport biomechanics and training science. Sensors (Basel). 2024, 24, 4616.
70. Isern-Kebschull, J.; Mechó, S.; Pedret, C.; et al. Muscle healing in sports injuries: MRI findings and proposed classification based on a single institutional experience and clinical observation. Radiographics 2024, 44, e230147.
71. Michaud, F.; Pérez Soto, M.; Lugrís, U.; Cuadrado, J. Lower back injury prevention and sensitization of hip hinge with neutral spine using wearable sensors during lifting exercises. Sensors (Basel). 2021, 21, 5487.
72. Assalve, G.; Lunetti, P.; Di Cagno, A.; et al. Advanced wearable devices for monitoring sweat biochemical markers in athletic performance: a comprehensive review. Biosensors 2024, 14, 574.
73. Alvarez, J. T.; Gerez, L. F.; Araromi, O. A.; et al. Toward soft wearable strain sensors for muscle activity monitoring. IEEE Trans. Neural Syst. Rehabil. Eng. 2022, 30, 2198-206.
74. Zhang, W.; Bai, Z.; Yan, P.; Liu, H.; Shao, L. Recognition of human lower limb motion and muscle fatigue status using a wearable FES-sEMG system. Sensors (Basel). 2024, 24, 2377.
75. Jia, W.; Bandodkar, A. J.; Valdés-Ramírez, G.; et al. Electrochemical tattoo biosensors for real-time noninvasive lactate monitoring in human perspiration. Anal. Chem. 2013, 85, 6553-60.
76. Anastasova, S.; Crewther, B.; Bembnowicz, P.; et al. A wearable multisensing patch for continuous sweat monitoring. Biosens. Bioelectron. 2017, 93, 139-45.
77. Meng, L.; Liu, S.; Borsa, B. A.; Eriksson, M.; Mak, W. C. A conducting polymer-based array with multiplex sensing and drug delivery capabilities for smart bandages. Commun. Mater. 2024, 5, 28.
78. Vaquer, A., Baron, E., de la Rica, R. Dissolvable polymer valves for sweat chrono-sampling in wearable paper-based analytical devices. ACS Sensors 2022, 7, 488-94.
79. Tardy, A. L.; Pouteau, E.; Marquez, D.; Yilmaz, C.; Scholey, A. Vitamins and minerals for energy, fatigue and cognition: a narrative review of the biochemical and clinical evidence. Nutrients 2020, 12, 228.
80. Zhu, Y.; Sun, S.; Yin, X.; Liu, Y.; Yang, W.; Chen, Y. Carbon nanotube-gold nanoparticle-based self-powered electrochemical biosensors for highly sensitive and stable detection of myoglobin. ACS Appl. Nano Mater. 2023, 6, 11085-94.
81. Lee, J.; Oh, Y.; Kim, H.; Lee, S.; Park, H. G. Personal glucose meter-based portable method for creatine kinase assay. Biosens. Bioelectron. X 2025, 22, 100575.
82. Elango, P. F. M.; Perera, G.; Yang, M.; et al. Multiplexed cutaneous wound monitor for point-of-care applications. Adv. NanoBiomed Res. 2025, 5, 2500142.
83. Giorgi-Coll, S.; Marín, M. J.; Sule, O.; Hutchinson, P. J.; Carpenter, K. L. H. Aptamer-modified gold nanoparticles for rapid aggregation-based detection of inflammation: an optical assay for interleukin-6. Mikrochim. Acta 2019, 187, 13.
84. Lee, S.; Ho, D. H.; Jekal, J.; et al. Fabric-based lamina emergent MXene-based electrode for electrophysiological monitoring. Nat. Commun. 2024, 15, 5974.
85. Hsu, Y.; Lu, Y.; Wang, T.; et al. An ultra-compliant P(VDF-TrFE) fiber-based muscle patch sensor for a new wearable application: monitoring muscle activities and fatigue through the peripheral length of a muscle’s physiological cross-sectional area. Sensor. Actuat. A-Phys. 2025, 393, 116823.
86. Song, E.; Xie, Z.; Bai, W.; et al. Miniaturized electromechanical devices for the characterization of the biomechanics of deep tissue. Nat. Biomed. Eng. 2021, 5, 759-71.
87. Yu, X.; Wang, H.; Ning, X.; et al. Needle-shaped ultrathin piezoelectric microsystem for guided tissue targeting via mechanical sensing. Nat. Biomed. Eng. 2018, 2, 165-72.
88. Liu, H. C.; Zeng, Y.; Gong, C.; et al. Wearable bioadhesive ultrasound shear wave elastography. Sci. Adv. 2024, 10, eadk8426.
89. Wang, Y.; Henderson, J.; Hafiz, P.; et al. Near-infrared spectroscopy-enabled electromechanical systems for fast mapping of biomechanics and subcutaneous diagnosis. Sci. Adv. 2024, 10, eadq9358.
90. Farina, D.; Enoka, R. M. Evolution of surface electromyography: from muscle electrophysiology towards neural recording and interfacing. J. Electromyogr. Kinesiol. 2023, 71, 102796.
91. Cheng, L.; Li, J.; Guo, A.; Zhang, J. Recent advances in flexible noninvasive electrodes for surface electromyography acquisition. NPJ Flex. Electron. 2023, 7, 39.
92. Tacca, N.; Dunlap, C.; Donegan, S. P.; et al. Wearable high-density EMG sleeve for complex hand gesture classification and continuous joint angle estimation. Sci. Rep. 2024, 14, 18564.
93. Meyers, E. C.; Gabrieli, D.; Tacca, N.; et al. Decoding hand and wrist movement intention from chronic stroke survivors with hemiparesis using a user-friendly, wearable EMG-based neural interface. J. Neuroeng. Rehabil. 2024, 21, 7.
94. Moin, A.; Zhou, A.; Rahimi, A.; et al. An EMG gesture recognition system with flexible high-density sensors and brain-inspired high-dimensional classifier. In 2018 IEEE International Symposium on Circuits and Systems (ISCAS), Florence, May 27-30, 2018; IEEE, 2018, pp 1-5.
95. Resnik, L.; Huang, H. H.; Winslow, A.; Crouch, D. L.; Zhang, F.; Wolk, N. Evaluation of EMG pattern recognition for upper limb prosthesis control: a case study in comparison with direct myoelectric control. J. Neuroeng. Rehabil. 2018, 15, 23.
96. Rea, M. S.; Nagare, R.; Figueiro, M. G. Modeling circadian phototransduction: quantitative predictions of psychophysical data. Front. Neurosci. 2021, 15, 615322.
97. Enoka, R. M.; Duchateau, J. Rate coding and the control of muscle force. Cold Spring Harb. Perspect. Med. 2017, 7, a029702.
98. Qiu, C.; Wu, F.; Han, W.; Yuce, M. R. A wearable bioimpedance chest patch for real-time ambulatory respiratory monitoring. IEEE Trans. Biomed. Eng. 2022, 69, 2970-81.
99. Stupin, D. D.; Kuzina, E. A.; Abelit, A. A.; et al. Bioimpedance spectroscopy: basics and applications. ACS Biomater. Sci. Eng. 2021, 7, 1962-86.
100. Bayoumy, K.; Gaber, M.; Elshafeey, A.; et al. Smart wearable devices in cardiovascular care: where we are and how to move forward. Nat. Rev. Cardiol. 2021, 18, 581-99.
101. Peabody, J. E.; Ryznar, R.; Ziesmann, M. T.; Gillman, L. A systematic review of heart rate variability as a measure of stress in medical professionals. Cureus 2023, 15, e34345.
102. Johnstone, J. A.; Ford, P. A.; Hughes, G.; Watson, T.; Garrett, A. T. Bioharness(TM) multivariable monitoring device: part. I: validity. J. Sports Sci. Med. 2012, 11, 400-8.
103. Khalili, M.; GholamHosseini, H.; Lowe, A.; Kuo, M. M. Y. Motion artifacts in capacitive ECG monitoring systems: a review of existing models and reduction techniques. Med. Biol. Eng. Comput. 2024, 62, 3599-622.
104. Van Doornik, D. M.; Berejikian, B. A.; Claiborne, A. M. Genetic monitoring of a long-term, large-scale experimental steelhead supplementation program. PLoS ONE 2025, 20, e0339458.
105. Pickart, L.; Margolina, A. Regenerative and protective actions of the GHK-Cu peptide in the light of the new gene data. Int. J. Mol. Sci. 2018, 19, 1987.
106. Brancaccio, A.; Tabarelli, D.; Bigica, M.; Baldauf, D. Cortical source localization of sleep-stage specific oscillatory activity. Sci. Rep. 2020, 10, 6976.
107. Zhong, S.; Jia, N.; Qu, Y.; et al. Analysis and study on biomarkers of local muscle fatigue caused by repetitive lifting task. BMC Musculoskelet. Disord. 2024, 25, 660.
108. Deng, Y. T.; You, J.; He, Y.; et al. Atlas of the plasma proteome in health and disease in 53,026 adults. Cell 2025, 188, 253-271.e7.
109. Gao, F.; Liu, C.; Zhang, L.; et al. Wearable and flexible electrochemical sensors for sweat analysis: a review. Microsyst. Nanoeng. 2023, 9, 1.
110. Evert, B.; Peterson, K. L.; Songkakul, T.; Bozkurt, A.; Daniele, M. A. Multimodal wearable platform with simultaneous electrochemical and biophotonic sensing. ACS Sens. 2025, 10, 5495-507.
111. Zhou, S.; Zhong, Q.; Wang, Y.; et al. Chemically engineered mesoporous silica nanoparticles-based intelligent delivery systems for theranostic applications in multiple cancerous/non-cancerous diseases. Coord. Chem. Rev. 2022, 452, 214309.
112. Wu, T.; Liu, G. Non-invasive wearables in inflammation monitoring: from biomarkers to biosensors. Biosensors 2025, 15, 351.
113. Bandodkar, A. J.; Jeang, W. J.; Ghaffari, R.; Rogers, J. A. wearable sensors for biochemical sweat analysis. Annu. Rev. Anal. Chem. (Palo Alto. Calif). 2019, 12, 1-22.
114. Ibáñez-Redín, G.; Rosso Cagnani, G.; O Gomes, N.; et al. Wearable potentiometric biosensor for analysis of urea in sweat. Biosens. Bioelectron. 2023, 223, 114994.
115. Chen, H.; Zhao, L.; Dong, W.; et al. Spatiotemporal variation of mortality burden attributable to heatwaves in China, 1979-2020. Sci. Bull. (Beijing). 2022, 67, 1340-4.
116. Mei, X.; Yang, J.; Liu, J.; Li, Y. Wearable, nanofiber-based microfluidic systems with integrated electrochemical and colorimetric sensing arrays for multiplex sweat analysis. Chem. Eng. J. 2023, 454, 140248.
117. Xiao, P.; Yun, X.; Chen, Y.; et al. Insights into the solvation chemistry in liquid electrolytes for lithium-based rechargeable batteries. Chem. Soc. Rev. 2023, 52, 5255-316.
118. Kim, G.; Ahn, H.; Chaj Ulloa, J.; Gao, W. Microneedle sensors for dermal interstitial fluid analysis. Med. X. 2024, 2, 15.
119. Hayes, E. J.; Stevenson, E.; Sayer, A. A.; Granic, A.; Hurst, C. Recovery from resistance exercise in older adults: a systematic scoping review. Sports Med. Open 2023, 9, 51.
120. Fishbein, A. B.; Knutson, K. L.; Zee, P. C. Circadian disruption and human health. J. Clin. Invest. 2021, 131, e148286.
121. Pan, X. F.; Wang, L.; Pan, A. Epidemiology and determinants of obesity in China. Lancet. Diabetes Endocrinol. 2021, 9, 373-92.
122. Wu, Z.; Qiao, Z.; Chen, S.; et al. Interstitial fluid-based wearable biosensors for minimally invasive healthcare and biomedical applications. Commun. Mater. 2024, 5, 33.
123. Stoffel, E. M.; Brand, R. E.; Goggins, M. Pancreatic cancer: changing epidemiology and new approaches to risk assessment, early detection, and prevention. Gastroenterology 2023, 164, 752-65.
124. Brancaccio, P.; Maffulli, N.; Limongelli, F. M. Creatine kinase monitoring in sport medicine. Br. Med. Bull. 2007, 81-82, 209-30.
125. Haller, N.; Behringer, M.; Reichel, T.; et al. Blood-based biomarkers for managing workload in athletes: considerations and recommendations for evidence-based use of established biomarkers. Sports Med. 2023, 53, 1315-33.
126. Wang, Z.; Luan, J.; Seth, A.; et al. Microneedle patch for the ultrasensitive quantification of protein biomarkers in interstitial fluid. Nat. Biomed. Eng. 2021, 5, 64-76.
127. Gao, Y.; Nguyen, D. T.; Yeo, T.; et al. A flexible multiplexed immunosensor for point-of-care in situ wound monitoring. Sci. Adv. 2021, 7.
128. Agustí, A.; Hogg, J. C. Update on the pathogenesis of chronic obstructive pulmonary disease. N. Engl. J. Med. 2019, 381, 1248-56.
129. Stark, T.; Walker, B.; Phillips, J. K.; Fejer, R.; Beck, R. Hand-held dynamometry correlation with the gold standard isokinetic dynamometry: a systematic review. PM R. 2011, 3, 472-9.
130. Huang, L.; Liu, Y.; Lin, T.; et al. Reliability and validity of two hand dynamometers when used by community-dwelling adults aged over 50 years. BMC Geriatr. 2022, 22, 580.
131. Garrett, S.; Jenkinson, T.; Kennedy, L. G.; Whitelock, H.; Gaisford, P.; Calin, A. A new approach to defining disease status in ankylosing spondylitis: the bath ankylosing spondylitis disease activity index. J. Rheumatol. 1994, 21, 2286-91.
132. Lang, S.; Yang, J.; Zhang, Y.; et al. Application of wearable insole sensors in in-place running: estimating lower limb load using machine learning. Biosensors 2025, 15, 83.
133. Knihs, D. A.; Zimmermann, H. B.; Pupo, J. D. Acute and delayed effects of fatigue on ground reaction force, lower limb stiffness and coordination asymmetries during a landing task. J. Hum. Kinet. 2021, 76, 191-9.
134. Sun, Q.; Yu, D.; Fan, J.; et al. China Kadoorie Biobank Collaborative Group. Healthy lifestyle and life expectancy at age 30 years in the Chinese population: an observational study. Lancet. Public Health 2022, 7, e994-e1004.
135. Tsuchida, W.; Yamakoshi, Y.; Matsuo, S.; et al. Application of the novel estimation method by shear wave elastography using vibrator to human skeletal muscle. Sci. Rep. 2020, 10, 22248.
136. Ohiri, K. A.; Pyles, C. O.; Hamilton, L. H.; et al. E-textile based modular sEMG suit for large area level of effort analysis. Sci. Rep. 2022, 12, 9650.
137. Pinelli, S.; Mandorino, M.; Lacome, M.; Fantozzi, S. Evaluation of WIMU sensor performance in estimating running stride and vertical stiffness in football training sessions: a comparison with smart insoles. Sensors (Basel). 2024, 24, 8087.
138. Oubre, B.; Lane, S.; Holmes, S.; Boyer, K.; Lee, S. I. Estimating ground reaction force and center of pressure using low-cost wearable devices. IEEE Trans. Biomed. Eng. 2022, 69, 1461-8.
139. Moraes, J. L.; Rocha, M. X.; Vasconcelos, G. G.; Vasconcelos Filho, J. E.; de Albuquerque, V. H. C.; Alexandria, A. R. Advances in photopletysmography signal analysis for biomedical applications. Sensors (Basel). 2018, 18, 1894.
140. Imani, S.; Bandodkar, A. J.; Mohan, A. M.; et al. A wearable chemical-electrophysiological hybrid biosensing system for real-time health and fitness monitoring. Nat. Commun. 2016, 7, 11650.
141. Resnik, L.; Huang, H.; Winslow, A.; Crouch, D. L.; Zhang, F.; Wolk, N. Evaluation of EMG pattern recognition for upper limb prosthesis control: a case study in comparison with direct myoelectric control. J. NeuroEngineering. Rehabil. 2018, 15, 23.
142. Wang, Z.; Fu, Y.; Bu, T.; et al. A soft on-skin sensor simultaneously capturing bioelectrical and biomechanical signals for muscle spasticity detection and assessment. Adv. Funct. Mater. 2025, 36, e23580.
143. Gao, W.; Emaminejad, S.; Nyein, H. Y. Y.; et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 2016, 529, 509-14.
144. He, X.; Fan, C.; Luo, Y.; Xu, T.; Zhang, X. Flexible microfluidic nanoplasmonic sensors for refreshable and portable recognition of sweat biochemical fingerprint. NPJ Flex. Electron. 2022, 6, 60.
145. Mascharak, S.; Griffin, M.; Talbott, H. E.; et al. Inhibiting mechanotransduction prevents scarring and yields regeneration in a large animal model. Sci. Transl. Med. 2025, 17, eadt6387.
146. McNamara, S. L.; Seo, B. R.; Freedman, B. R.; et al. Anti-inflammatory therapy enables robot-actuated regeneration of aged muscle. Sci. Robot. 2023, 8, eadd9369.
147. Alisafaei, F.; Shakiba, D.; Hong, Y.; et al. Tension anisotropy drives fibroblast phenotypic transition by self-reinforcing cell-extracellular matrix mechanical feedback. Nat. Mater. 2025, 24, 955-65.
148. Chen, J.; Wang, X.; Li, Q.; Shen, B.; Tao, X. Wearable multimodal sensing platform for contraction and fatigue transition monitoring of biceps in motion using machine learning. Sensor. Actuat. A-Phys. 2025, 396, 117127.
149. Westerblad, H.; Bruton, J. D.; Katz, A. Skeletal muscle: energy metabolism, fiber types, fatigue and adaptability. Exp. Cell Res. 2010, 316, 3093-9.
150. Hostrup, M.; Deshmukh, A. S. Fiber type-specific adaptations to exercise training in human skeletal muscle: lessons from proteome analyses and future directions. Scand. J. Med. Sci. Sports 2025, 35, e70059.
151. Sempionatto, J. R.; Montiel, V. R.; Vargas, E.; Teymourian, H.; Wang, J. Wearable and Mobile Sensors for Personalized Nutrition. ACS Sens. 2021, 6, 1745-60.
152. Yu, D.; Zhu, Z.; Sheng, Q.; et al. Multimodal wearable sensing for biomechanics and biomolecules enabled by the M-MPM/VCFs@Ag interface with machine learning pipeline. ACS Sens. 2025, 10, 4307-17.
153. S. Bai, J. Z. Kolter, V. J. A. Koltun, An empirical evaluation of generic convolutional and recurrent networks for sequence modeling. arXiv 2018;arXiv:1803.01271. Available online: https://doi.org/10.48550/arXiv.1803.01271. [accessed 13 April 2026].
154. Foltynie, T. Scaling up GMP-grade dopaminergic cells for Parkinson’s disease. Cell Stem Cell 2024, 31, 5-6.
155. Bradford, J. C.; Tweedell, A.; Leahy, L. High-density surface and intramuscular EMG data from the tibialis anterior during dynamic contractions. Sci. Data 2023, 10, 434.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data






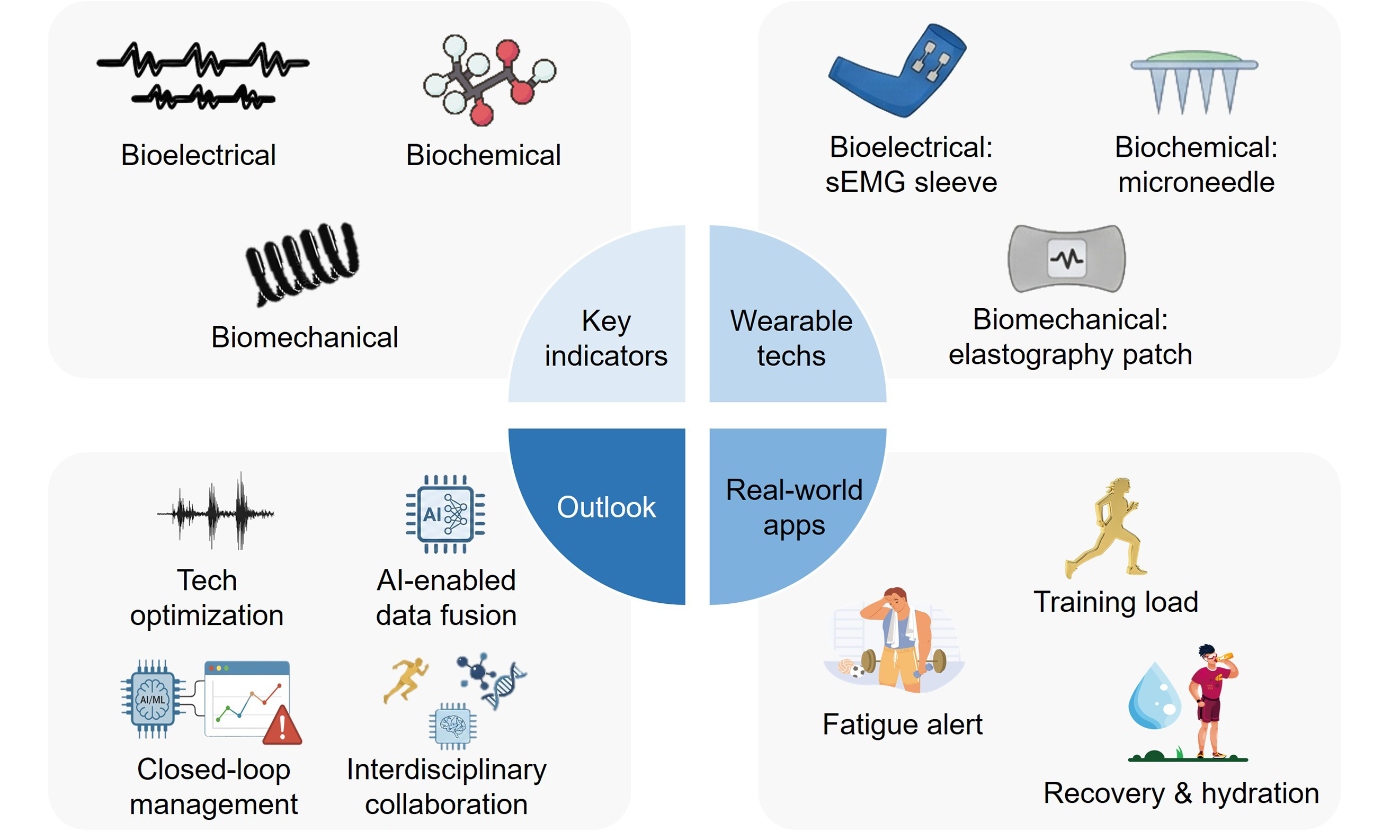
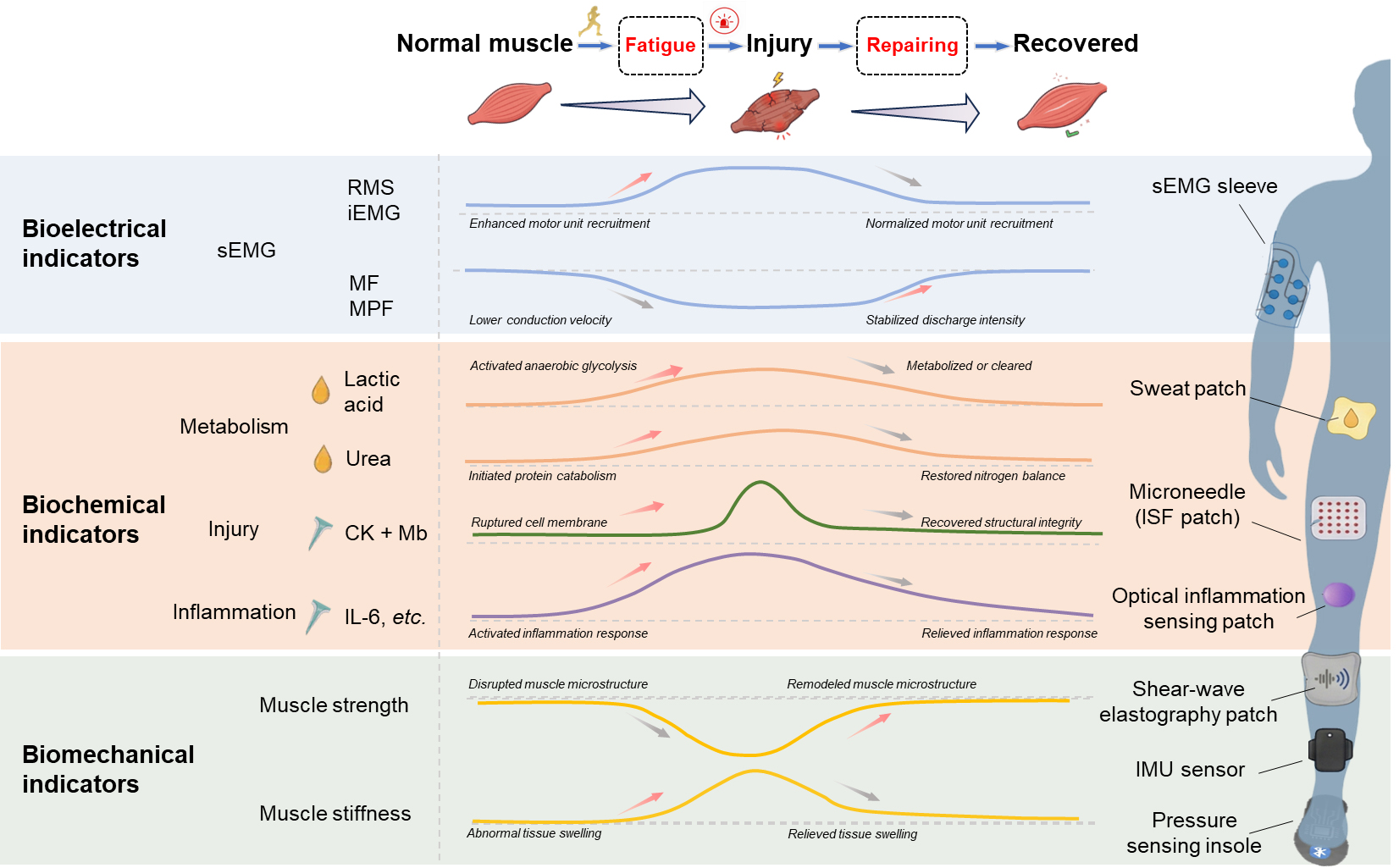
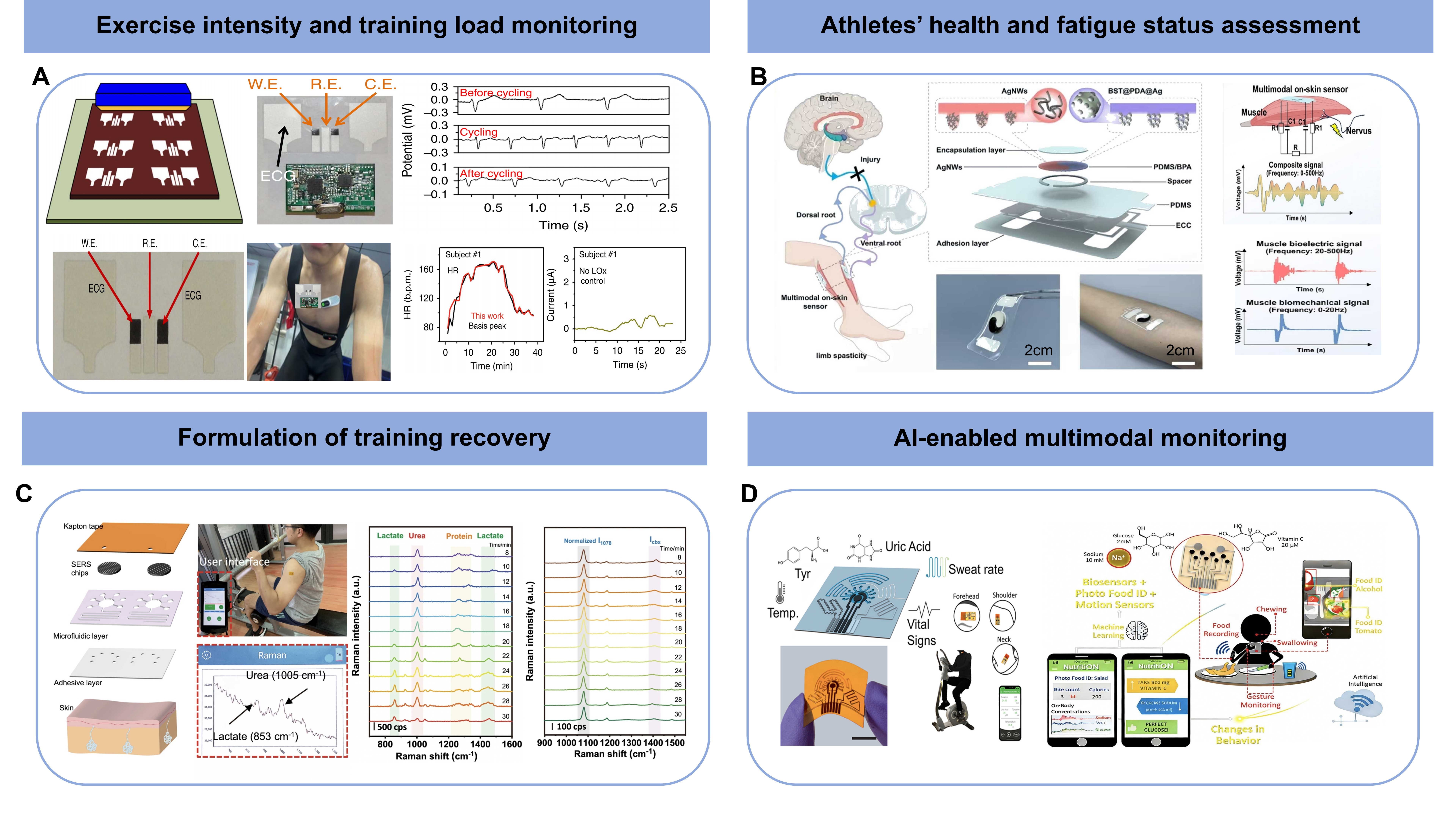
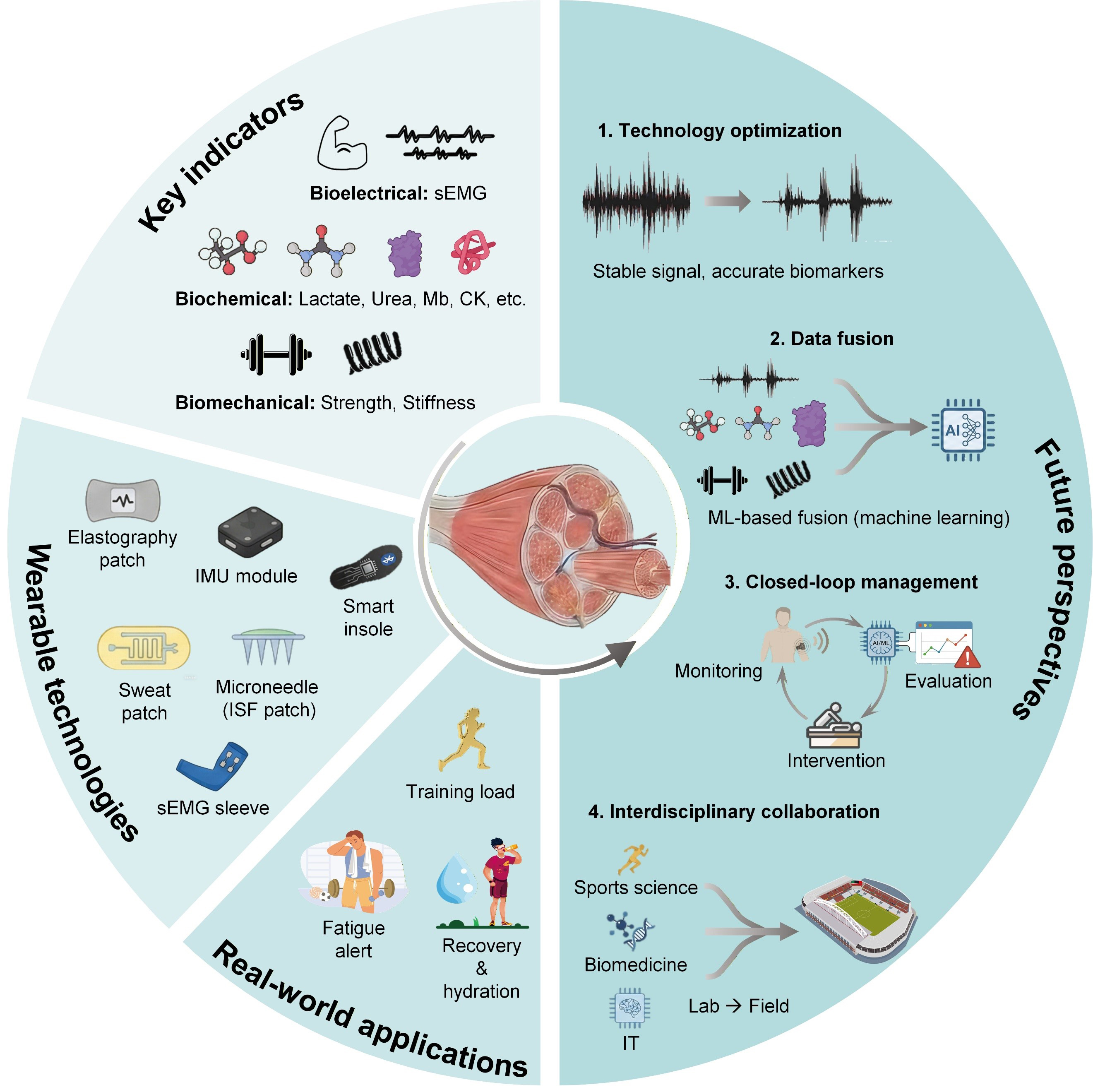












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at [email protected].