fig1
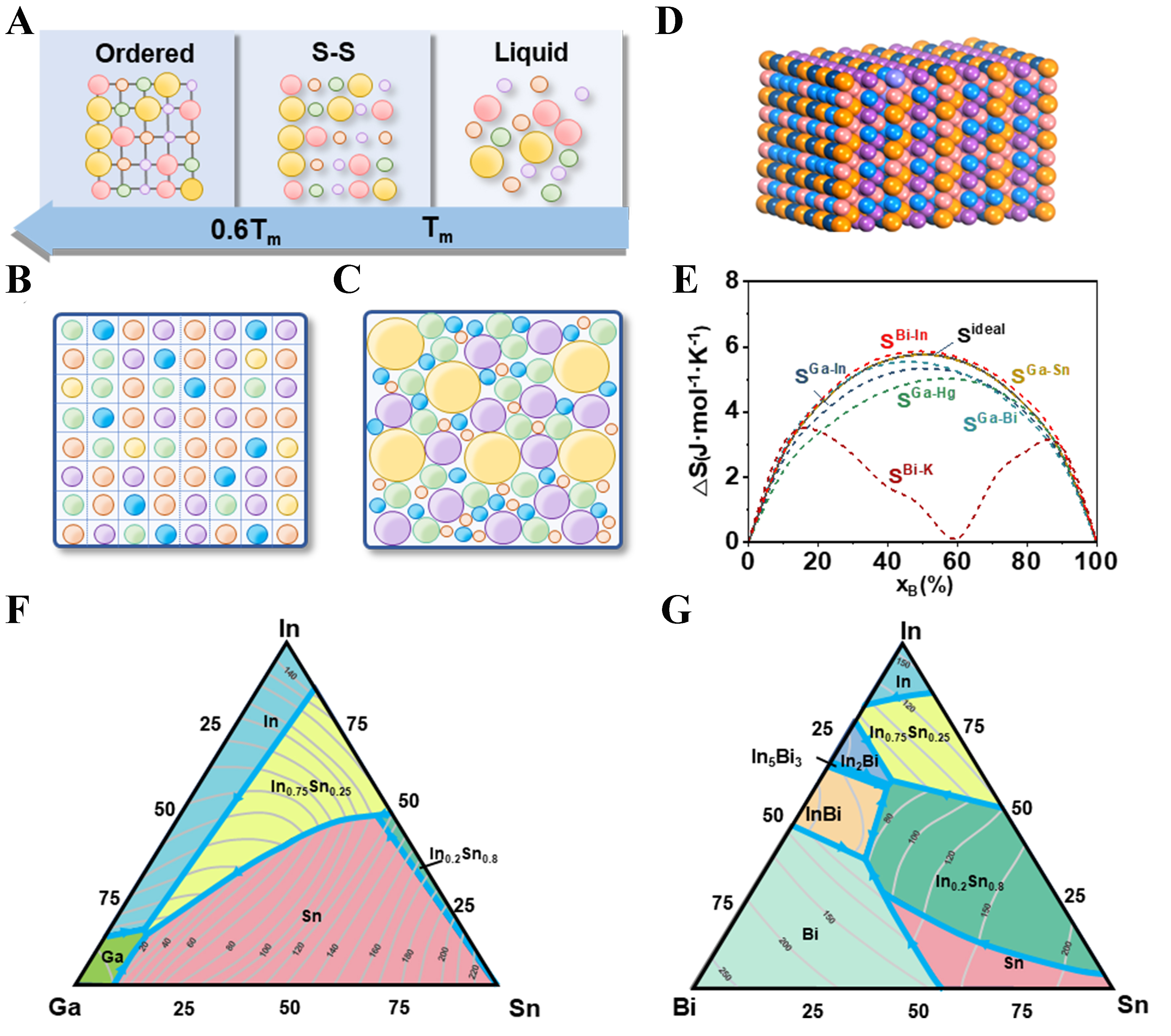
Figure 1. (A) Solidification process in a typical HEA, showing transition from liquid to solid solution and then to a multiphase disordered solid due to segregation and ordering. S-S represents the solid solution phase. Tm is the melting point; (B) Ideal atomic configuration with equal-sized atoms arranged in a regular lattice structure[6]; (C) Real atomic configuration in HEAs with varied atomic sizes, leading to random packing and increased entropy[6]; (D) 3D visualization of atom distribution in HEAs, showing disordered arrangements that contribute to excess entropy; (E) Comparison of phase behaviors in Ga-In and Bi-K systems, illustrating different entropy trends due to IMC formation. SA-B represents the total molar entropy ST of different binary systems; (F) Phase diagram of the Ga-In-Sn ternary alloy system with varying compositions and temperatures[7]; (G) Phase diagram of the Bi-In-Sn ternary alloy system[7]. (B and C) Adapted with permission from Ref.[6]. Copyright 2014 Elsevier. (F and G) Adapted with permission from Ref.[7]. Copyright 2001 Elsevier. HEA: High-entropy alloy; 3D: three-dimensional; IMC: intermetallic compound.